| Record Information |
|---|
| Version | 2.0 |
|---|
| Creation Date | 2009-03-06 18:58:06 UTC |
|---|
| Update Date | 2014-12-24 20:21:08 UTC |
|---|
| Accession Number | T3D0112 |
|---|
| Identification |
|---|
| Common Name | n-Nitrosodi-n-propylamine |
|---|
| Class | Small Molecule |
|---|
| Description | N-Nitrosodi-n-propylamine is a chemical produced by industry in small amounts for research. Small amounts of n-nitrosodi-n-propylamine are also produced as a side reaction during some manufacturing processes, as a contaminant in some weed killers, and during the manufacture of some rubber products. It is one ingredient of cigarette. (5) |
|---|
| Compound Type | - Amine
- Cigarette Toxin
- Industrial By-product/Pollutant
- Industrial/Workplace Toxin
- Nitrite
- Organic Compound
- Pollutant
- Synthetic Compound
|
|---|
| Chemical Structure | |
|---|
| Synonyms | | Synonym | | 2-Oxo-1,1-dipropylhydrazine | | Di-N-propylnitrosamine | | Di-N-propylnitrosoamine | | Dipropylnitrosamine | | N,N-di-N-propylnitrosamine | | N,N-dipropylnitrosamine | | N-nitroso di-N-propylamine | | N-nitroso(di-N-propyl)amine | | N-nitroso-N-dipropylamine | | N-Nitroso-N-propyl-1-propanamine | | N-nitroso-N-propylpropan-1-amine | | N-Nitrosodi-N-propylamine | | N-nitrosodi-N-propylamine | | N-nitrosodipropylamine | | Nitrosodipropylamine | | Nitrous dipropylamide |
|
|---|
| Chemical Formula | C6H14N2O |
|---|
| Average Molecular Mass | 130.188 g/mol |
|---|
| Monoisotopic Mass | 130.111 g/mol |
|---|
| CAS Registry Number | 621-64-7 |
|---|
| IUPAC Name | nitrosodipropylamine |
|---|
| Traditional Name | N-nitrosodipropylamine |
|---|
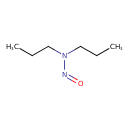
| SMILES | CCCN(CCC)N=O |
|---|
| InChI Identifier | InChI=1S/C6H14N2O/c1-3-5-8(7-9)6-4-2/h3-6H2,1-2H3 |
|---|
| InChI Key | InChIKey=YLKFDHTUAUWZPQ-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as organic n-nitroso compounds. These are organic compounds containing a n-nitroso group -NN=O. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic nitrogen compounds |
|---|
| Class | Organonitrogen compounds |
|---|
| Sub Class | Organic nitroso compounds |
|---|
| Direct Parent | Organic N-nitroso compounds |
|---|
| Alternative Parents | |
|---|
| Substituents | - Organic n-nitroso compound
- Organic oxygen compound
- Organopnictogen compound
- Organic oxide
- Hydrocarbon derivative
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Exogenous |
|---|
| Cellular Locations | |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Appearance | Yellow liquid. |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | 206 °C | | Solubility | 13 mg/mL at 24 °C [MIRVISH,SS et al.(1976)] | | LogP | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | Deposition Date | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0f6x-9200000000-7a59226c684fdfa7f3f4 | 2021-09-23 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 30V, Positive | splash10-0019-9500000000-0704dc1a231c669f02cb | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 45V, Positive | splash10-000i-9200000000-154023f596f82ff60612 | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 75V, Positive | splash10-0udr-9000000000-cf38c049fad6b029abf9 | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 60V, Positive | splash10-000i-9000000000-7f3e7dfc90998dac6805 | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 90V, Positive | splash10-0udi-9000000000-7817e43d165c2c3db63a | 2021-09-20 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-3900000000-519b13d74f5fd41dae33 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0006-9200000000-ec2e51e590dd7bb0651d | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0006-9000000000-b184a59d7647d7a9126a | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-1900000000-de2892f0b560af379291 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-01t9-4900000000-24bf802b1bb215f4745f | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9000000000-d063bf39130b38e25ac9 | 2016-08-03 | View Spectrum | | MS | Mass Spectrum (Electron Ionization) | splash10-0006-9000000000-632355cddcea307ac9e7 | 2014-09-20 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | Not Available | 2021-10-25 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | Not Available | 2021-10-25 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | Not Available | 2021-10-25 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | Not Available | 2021-10-25 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | Not Available | 2021-10-25 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | Not Available | 2021-10-25 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | Not Available | 2021-10-25 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | Not Available | 2021-10-25 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | Not Available | 2021-10-25 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | Not Available | 2021-10-25 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | Not Available | 2021-10-25 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | Not Available | 2021-10-25 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | Not Available | 2021-10-25 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | Not Available | 2021-10-25 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | Not Available | 2021-10-25 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | Not Available | 2021-10-25 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | Not Available | 2021-10-25 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | Not Available | 2021-10-25 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | Not Available | 2021-10-25 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | Not Available | 2021-10-25 | View Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Oral (5); inhalation (5) ; dermal (5). |
|---|
| Mechanism of Toxicity | Reactive metabolites of n-nitrosodi-n-propylamine are believed to form adducts with DNA, resulting in carcinogenic effects. (5) |
|---|
| Metabolism | n-Nitrosodi-n-propylamine can be absorbed through oral, inhalation, or dermal routes. It is metabolized by cytochrome p-450 enzymes (mainly CYP 2E1) into its reactive metabolites via oxidation at the alpha, beta and gamma carbon positions. Alpha carbon oxidation is regarded as the primary pathway, resulting in formation of propionaldehyde, 1-propanol, and 2-propanol. The metabolites of n-nitrosodi-n-propylamine are excreted mainly in the urine. (5, 1) |
|---|
| Toxicity Values | LD50: 480 mg/kg (Oral, Rat) (2)
LD50: 487 mg/kg (Subcutaneous, Rat) (2) |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | 2B, possibly carcinogenic to humans. (4) |
|---|
| Uses/Sources | Small amounts of n-nitrosodi-n-propylamine are also produced as a side reaction during some manufacturing processes, as a contaminant in some weed killers, and during the manufacture of some rubber products. (5) |
|---|
| Minimum Risk Level | Acute Oral: 0.095 mg/kg/day (3) |
|---|
| Health Effects | High levels of n-nitrosodi-n-propylamine may damage the liver, lung, stomach, kidneys, and heart. n-Nitrosodi-n-propylamine is also a likely carcinogen. (5) |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Normal Concentrations |
|---|
| Not Available |
|---|
| Abnormal Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| PubChem Compound ID | 12130 |
|---|
| ChEMBL ID | CHEMBL166242 |
|---|
| ChemSpider ID | 11632 |
|---|
| KEGG ID | C19279 |
|---|
| UniProt ID | Not Available |
|---|
| OMIM ID | |
|---|
| ChEBI ID | Not Available |
|---|
| BioCyc ID | CPD-630 |
|---|
| CTD ID | C013161 |
|---|
| Stitch ID | n-Nitrosodi-n-propylamine |
|---|
| PDB ID | Not Available |
|---|
| ACToR ID | 1053 |
|---|
| Wikipedia Link | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | T3D0112.pdf |
|---|
| General References | - Teiber JF, Hollenberg PF: Identification of the human liver microsomal cytochrome P450s involved in the metabolism of N-nitrosodi-n-propylamine. Carcinogenesis. 2000 Aug;21(8):1559-66. [10910959 ]
- IARC (1983). Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Man. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-Present. (Multivolume work).
- ATSDR - Agency for Toxic Substances and Disease Registry (2001). Minimal Risk Levels (MRLs) for Hazardous Substances. U.S. Public Health Service in collaboration with U.S. Environmental Protection Agency (EPA). [Link]
- International Agency for Research on Cancer (2014). IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. [Link]
- ATSDR - Agency for Toxic Substances and Disease Registry (1989). Toxicological profile for n-nitrosodi-n-propylamine. U.S. Public Health Service in collaboration with U.S. Environmental Protection Agency (EPA). [Link]
|
|---|
| Gene Regulation |
|---|
| Up-Regulated Genes | Not Available |
|---|
| Down-Regulated Genes | Not Available |
|---|