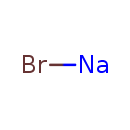Sodium bromide (T3D1754)
| Record Information | ||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 2.0 | |||||||||||||||||||||||||||||||||||||||||||||
| Creation Date | 2009-06-22 16:08:33 UTC | |||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 2014-12-24 20:24:35 UTC | |||||||||||||||||||||||||||||||||||||||||||||
| Accession Number | T3D1754 | |||||||||||||||||||||||||||||||||||||||||||||
| Identification | ||||||||||||||||||||||||||||||||||||||||||||||
| Common Name | Sodium bromide | |||||||||||||||||||||||||||||||||||||||||||||
| Class | Small Molecule | |||||||||||||||||||||||||||||||||||||||||||||
| Description | Sodium bromide is a chemical compound of sodium and bromine. It was widely used as an anticonvulsant and a sedative in the late 19th and early 20th centuries, but today is only used in veterinary medicine, as an antiepileptic medication for dogs and cats. It is also used in photography. Bromine is a halogen element with the symbol Br and atomic number 35. Diatomic bromine does not occur naturally, but bromine salts can be found in crustal rock. (3, 6) | |||||||||||||||||||||||||||||||||||||||||||||
| Compound Type |
| |||||||||||||||||||||||||||||||||||||||||||||
| Chemical Structure | ||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms |
| |||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula | BrNa | |||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Mass | 102.894 g/mol | |||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Mass | 101.908 g/mol | |||||||||||||||||||||||||||||||||||||||||||||
| CAS Registry Number | 7647-15-6 | |||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name | bromosodium | |||||||||||||||||||||||||||||||||||||||||||||
| Traditional Name | bromosodium | |||||||||||||||||||||||||||||||||||||||||||||
| SMILES | [Na]Br | |||||||||||||||||||||||||||||||||||||||||||||
| InChI Identifier | InChI=1S/BrH.Na/h1H;/q;+1/p-1 | |||||||||||||||||||||||||||||||||||||||||||||
| InChI Key | InChIKey=JHJLBTNAGRQEKS-UHFFFAOYSA-M | |||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | ||||||||||||||||||||||||||||||||||||||||||||||
| Description | belongs to the class of inorganic compounds known as alkali metal bromides. These are inorganic compounds in which the largest halogen atom is Bromine, and the heaviest metal atom an alkali metal. | |||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Inorganic compounds | |||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Mixed metal/non-metal compounds | |||||||||||||||||||||||||||||||||||||||||||||
| Class | Alkali metal salts | |||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Alkali metal bromides | |||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Alkali metal bromides | |||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | ||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| |||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors |
| |||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | ||||||||||||||||||||||||||||||||||||||||||||||
| Status | Detected and Not Quantified | |||||||||||||||||||||||||||||||||||||||||||||
| Origin | Exogenous | |||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations |
| |||||||||||||||||||||||||||||||||||||||||||||
| Biofluid Locations | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Tissue Locations | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Pathways | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Applications | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Biological Roles | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Chemical Roles | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | ||||||||||||||||||||||||||||||||||||||||||||||
| State | Solid | |||||||||||||||||||||||||||||||||||||||||||||
| Appearance | White powder. | |||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties |
| |||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| |||||||||||||||||||||||||||||||||||||||||||||
| Spectra | ||||||||||||||||||||||||||||||||||||||||||||||
| Spectra |
| |||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Profile | ||||||||||||||||||||||||||||||||||||||||||||||
| Route of Exposure | Oral (4) ; inhalation (4) ; dermal (4) | |||||||||||||||||||||||||||||||||||||||||||||
| Mechanism of Toxicity | Bromine is a powerful oxidizing agent and is able to release oxygen free radicals from the water in mucous membranes. These free radicals are also potent oxidizers and produce tissue damage. In additon, the formation of hydrobromic and bromic acids will result in secondary irritation. The bromide ion is also known to affect the central nervous system, causing bromism. This is believed to be a result of bromide ions substituting for chloride ions in the in actions of neurotransmitters and transport systems, thus affecting numerous synaptic processes. (4, 5, 1) | |||||||||||||||||||||||||||||||||||||||||||||
| Metabolism | Bromine is mainly absorbed via inhalation, but may also enter the body through dermal contact. Bromine salts can be ingested. Due to its reactivity, bromine quickly forms bromide and may be deposited in the tissues, displacing other halogens. (4) | |||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Values | LD50: 3500 mg/kg (Oral, Rat) (7) LD50: 2900 mg/kg (Subcutaneous, Rat) (7) LD50: 5000 mg/kg (Intraperitoneal, Mouse) (7) | |||||||||||||||||||||||||||||||||||||||||||||
| Lethal Dose | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Carcinogenicity (IARC Classification) | No indication of carcinogenicity to humans (not listed by IARC). | |||||||||||||||||||||||||||||||||||||||||||||
| Uses/Sources | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Minimum Risk Level | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Health Effects | Bromine vapour causes irritation and direct damage to the mucous membranes. Elemental bromine also burns the skin. The bromide ion is a central nervous system depressant and chronic exposure produces neuronal effects. This is called bromism and can result in central reactions reaching from somnolence to coma, cachexia, exicosis, loss of reflexes or pathologic reflexes, clonic seizures, tremor, ataxia, loss of neural sensitivity, paresis, papillar edema of the eyes, abnormal speech, cerebral edema, delirium, aggressiveness, and psychoses. (3, 4, 5) | |||||||||||||||||||||||||||||||||||||||||||||
| Symptoms | Bromine vapour causes irritation and direct damage to the mucous membranes. Symptoms include lacrimation, rhinorrhoea, eye irritation with mucous secretions from the oropharyngeal and upper airways, coughing, dyspnoea, choking, wheezing, epistaxis, and headache. The bromide ion is a central nervous system depressant producing ataxia, slurred speech, tremor, nausea, vomiting, lethargy, dizziness, visual disturbances, unsteadiness, headaches, impaired memory and concentration, disorientation and hallucinations. This is called bromism. (4, 5) | |||||||||||||||||||||||||||||||||||||||||||||
| Treatment | EYES: irrigate opened eyes for several minutes under running water. INGESTION: do not induce vomiting. Rinse mouth with water (never give anything by mouth to an unconscious person). Seek immediate medical advice. SKIN: should be treated immediately by rinsing the affected parts in cold running water for at least 15 minutes, followed by thorough washing with soap and water. If necessary, the person should shower and change contaminated clothing and shoes, and then must seek medical attention. INHALATION: supply fresh air. If required provide artificial respiration. | |||||||||||||||||||||||||||||||||||||||||||||
| Normal Concentrations | ||||||||||||||||||||||||||||||||||||||||||||||
| Not Available | ||||||||||||||||||||||||||||||||||||||||||||||
| Abnormal Concentrations | ||||||||||||||||||||||||||||||||||||||||||||||
| Not Available | ||||||||||||||||||||||||||||||||||||||||||||||
| External Links | ||||||||||||||||||||||||||||||||||||||||||||||
| DrugBank ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| HMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| PubChem Compound ID | 253881 | |||||||||||||||||||||||||||||||||||||||||||||
| ChEMBL ID | CHEMBL1644694 | |||||||||||||||||||||||||||||||||||||||||||||
| ChemSpider ID | 22712 | |||||||||||||||||||||||||||||||||||||||||||||
| KEGG ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| UniProt ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| OMIM ID | ||||||||||||||||||||||||||||||||||||||||||||||
| ChEBI ID | 63004 | |||||||||||||||||||||||||||||||||||||||||||||
| BioCyc ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| CTD ID | C027938 | |||||||||||||||||||||||||||||||||||||||||||||
| Stitch ID | Sodium bromide | |||||||||||||||||||||||||||||||||||||||||||||
| PDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| ACToR ID | 12136 | |||||||||||||||||||||||||||||||||||||||||||||
| Wikipedia Link | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| References | ||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| MSDS | T3D1754.pdf | |||||||||||||||||||||||||||||||||||||||||||||
| General References |
| |||||||||||||||||||||||||||||||||||||||||||||
| Gene Regulation | ||||||||||||||||||||||||||||||||||||||||||||||
| Up-Regulated Genes | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Down-Regulated Genes | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
Targets
- General Function:
- Zinc ion binding
- Specific Function:
- Essential for bone resorption and osteoclast differentiation (By similarity). Reversible hydration of carbon dioxide. Can hydrate cyanamide to urea. Involved in the regulation of fluid secretion into the anterior chamber of the eye. Contributes to intracellular pH regulation in the duodenal upper villous epithelium during proton-coupled peptide absorption. Stimulates the chloride-bicarbonate exchange activity of SLC26A6.
- Gene Name:
- CA2
- Uniprot ID:
- P00918
- Molecular Weight:
- 29245.895 Da
Binding/Activity Constants
| Type | Value | Assay Type | Assay Source |
|---|---|---|---|
| Inhibitory | 63000 uM | Not Available | BindingDB 50339814 |
References
- Cincinelli A, Martellini T, Innocenti A, Scozzafava A, Supuran CT: Purification and inhibition studies with anions and sulfonamides of an alpha-carbonic anhydrase from the Antarctic seal Leptonychotes weddellii. Bioorg Med Chem. 2011 Mar 15;19(6):1847-51. doi: 10.1016/j.bmc.2011.02.015. Epub 2011 Feb 13. [21377369 ]
- Vullo D, Nishimori I, Minakuchi T, Scozzafava A, Supuran CT: Inhibition studies with anions and small molecules of two novel beta-carbonic anhydrases from the bacterial pathogen Salmonella enterica serovar Typhimurium. Bioorg Med Chem Lett. 2011 Jun 15;21(12):3591-5. doi: 10.1016/j.bmcl.2011.04.105. Epub 2011 Apr 28. [21570835 ]
- Vullo D, De Luca V, Scozzafava A, Carginale V, Rossi M, Supuran CT, Capasso C: Anion inhibition studies of the fastest carbonic anhydrase (CA) known, the extremo-CA from the bacterium Sulfurihydrogenibium azorense. Bioorg Med Chem Lett. 2012 Dec 1;22(23):7142-5. doi: 10.1016/j.bmcl.2012.09.065. Epub 2012 Sep 27. [23072866 ]
- Nishimori I, Vullo D, Minakuchi T, Scozzafava A, Capasso C, Supuran CT: Restoring catalytic activity to the human carbonic anhydrase (CA) related proteins VIII, X and XI affords isoforms with high catalytic efficiency and susceptibility to anion inhibition. Bioorg Med Chem Lett. 2013 Jan 1;23(1):256-60. doi: 10.1016/j.bmcl.2012.10.103. Epub 2012 Nov 9. [23200251 ]
- Monti SM, De Simone G, Dathan NA, Ludwig M, Vullo D, Scozzafava A, Capasso C, Supuran CT: Kinetic and anion inhibition studies of a beta-carbonic anhydrase (FbiCA 1) from the C4 plant Flaveria bidentis. Bioorg Med Chem Lett. 2013 Mar 15;23(6):1626-30. doi: 10.1016/j.bmcl.2013.01.087. Epub 2013 Jan 30. [23414801 ]
- Vullo D, Isik S, Del Prete S, De Luca V, Carginale V, Scozzafava A, Supuran CT, Capasso C: Anion inhibition studies of the alpha-carbonic anhydrase from the pathogenic bacterium Vibrio cholerae. Bioorg Med Chem Lett. 2013 Mar 15;23(6):1636-8. doi: 10.1016/j.bmcl.2013.01.084. Epub 2013 Jan 29. [23414807 ]
- General Function:
- Zinc ion binding
- Specific Function:
- Reversible hydration of carbon dioxide. Can hydrates cyanamide to urea.
- Gene Name:
- CA1
- Uniprot ID:
- P00915
- Molecular Weight:
- 28870.0 Da
Binding/Activity Constants
| Type | Value | Assay Type | Assay Source |
|---|---|---|---|
| Inhibitory | 4000 uM | Not Available | BindingDB 50339814 |
References
- Cincinelli A, Martellini T, Innocenti A, Scozzafava A, Supuran CT: Purification and inhibition studies with anions and sulfonamides of an alpha-carbonic anhydrase from the Antarctic seal Leptonychotes weddellii. Bioorg Med Chem. 2011 Mar 15;19(6):1847-51. doi: 10.1016/j.bmc.2011.02.015. Epub 2011 Feb 13. [21377369 ]
- Vullo D, Nishimori I, Minakuchi T, Scozzafava A, Supuran CT: Inhibition studies with anions and small molecules of two novel beta-carbonic anhydrases from the bacterial pathogen Salmonella enterica serovar Typhimurium. Bioorg Med Chem Lett. 2011 Jun 15;21(12):3591-5. doi: 10.1016/j.bmcl.2011.04.105. Epub 2011 Apr 28. [21570835 ]
- Vullo D, De Luca V, Scozzafava A, Carginale V, Rossi M, Supuran CT, Capasso C: Anion inhibition studies of the fastest carbonic anhydrase (CA) known, the extremo-CA from the bacterium Sulfurihydrogenibium azorense. Bioorg Med Chem Lett. 2012 Dec 1;22(23):7142-5. doi: 10.1016/j.bmcl.2012.09.065. Epub 2012 Sep 27. [23072866 ]
- Nishimori I, Vullo D, Minakuchi T, Scozzafava A, Capasso C, Supuran CT: Restoring catalytic activity to the human carbonic anhydrase (CA) related proteins VIII, X and XI affords isoforms with high catalytic efficiency and susceptibility to anion inhibition. Bioorg Med Chem Lett. 2013 Jan 1;23(1):256-60. doi: 10.1016/j.bmcl.2012.10.103. Epub 2012 Nov 9. [23200251 ]
- Vullo D, Isik S, Del Prete S, De Luca V, Carginale V, Scozzafava A, Supuran CT, Capasso C: Anion inhibition studies of the alpha-carbonic anhydrase from the pathogenic bacterium Vibrio cholerae. Bioorg Med Chem Lett. 2013 Mar 15;23(6):1636-8. doi: 10.1016/j.bmcl.2013.01.084. Epub 2013 Jan 29. [23414807 ]
- General Function:
- Voltage-gated chloride channel activity
- Specific Function:
- Voltage-gated chloride channel. Chloride channels have several functions including the regulation of cell volume; membrane potential stabilization, signal transduction and transepithelial transport.
- Gene Name:
- CLCN1
- Uniprot ID:
- P35523
- Molecular Weight:
- 108625.435 Da
References
- Simchowitz L: Interactions of bromide, iodide, and fluoride with the pathways of chloride transport and diffusion in human neutrophils. J Gen Physiol. 1988 Jun;91(6):835-60. [3047312 ]
- Pusch M, Jordt SE, Stein V, Jentsch TJ: Chloride dependence of hyperpolarization-activated chloride channel gates. J Physiol. 1999 Mar 1;515 ( Pt 2):341-53. [10050002 ]
- General Function:
- Voltage-gated chloride channel activity
- Specific Function:
- Voltage-gated chloride channel. Chloride channels have several functions including the regulation of cell volume; membrane potential stabilization, signal transduction and transepithelial transport. May be important in urinary concentrating mechanisms.
- Gene Name:
- CLCNKA
- Uniprot ID:
- P51800
- Molecular Weight:
- 75284.08 Da
References
- Simchowitz L: Interactions of bromide, iodide, and fluoride with the pathways of chloride transport and diffusion in human neutrophils. J Gen Physiol. 1988 Jun;91(6):835-60. [3047312 ]
- Pusch M, Jordt SE, Stein V, Jentsch TJ: Chloride dependence of hyperpolarization-activated chloride channel gates. J Physiol. 1999 Mar 1;515 ( Pt 2):341-53. [10050002 ]
- General Function:
- Voltage-gated chloride channel activity
- Specific Function:
- Voltage-gated chloride channel. Chloride channels have several functions including the regulation of cell volume; membrane potential stabilization, signal transduction and transepithelial transport. May be important in urinary concentrating mechanisms.
- Gene Name:
- CLCNKB
- Uniprot ID:
- P51801
- Molecular Weight:
- 75445.3 Da
References
- Simchowitz L: Interactions of bromide, iodide, and fluoride with the pathways of chloride transport and diffusion in human neutrophils. J Gen Physiol. 1988 Jun;91(6):835-60. [3047312 ]
- Pusch M, Jordt SE, Stein V, Jentsch TJ: Chloride dependence of hyperpolarization-activated chloride channel gates. J Physiol. 1999 Mar 1;515 ( Pt 2):341-53. [10050002 ]
- General Function:
- Inhibitory extracellular ligand-gated ion channel activity
- Specific Function:
- Component of the heteropentameric receptor for GABA, the major inhibitory neurotransmitter in the vertebrate brain. Functions also as histamine receptor and mediates cellular responses to histamine. Functions as receptor for diazepines and various anesthetics, such as pentobarbital; these are bound at a separate allosteric effector binding site. Functions as ligand-gated chloride channel (By similarity).
- Gene Name:
- GABRA1
- Uniprot ID:
- P14867
- Molecular Weight:
- 51801.395 Da
References
- Suzuki S, Kawakami K, Nakamura F, Nishimura S, Yagi K, Seino M: Bromide, in the therapeutic concentration, enhances GABA-activated currents in cultured neurons of rat cerebral cortex. Epilepsy Res. 1994 Oct;19(2):89-97. [7843172 ]
- General Function:
- Gaba-gated chloride ion channel activity
- Specific Function:
- Component of the heteropentameric receptor for GABA, the major inhibitory neurotransmitter in the vertebrate brain. Functions also as histamine receptor and mediates cellular responses to histamine. Functions as receptor for diazepines and various anesthetics, such as pentobarbital; these are bound at a separate allosteric effector binding site. Functions as ligand-gated chloride channel.
- Gene Name:
- GABRB3
- Uniprot ID:
- P28472
- Molecular Weight:
- 54115.04 Da
References
- Suzuki S, Kawakami K, Nakamura F, Nishimura S, Yagi K, Seino M: Bromide, in the therapeutic concentration, enhances GABA-activated currents in cultured neurons of rat cerebral cortex. Epilepsy Res. 1994 Oct;19(2):89-97. [7843172 ]
- General Function:
- Inhibitory extracellular ligand-gated ion channel activity
- Specific Function:
- Component of the heteropentameric receptor for GABA, the major inhibitory neurotransmitter in the vertebrate brain. Functions also as histamine receptor and mediates cellular responses to histamine. Functions as receptor for diazepines and various anesthetics, such as pentobarbital; these are bound at a separate allosteric effector binding site. Functions as ligand-gated chloride channel.
- Gene Name:
- GABRG2
- Uniprot ID:
- P18507
- Molecular Weight:
- 54161.78 Da
References
- Suzuki S, Kawakami K, Nakamura F, Nishimura S, Yagi K, Seino M: Bromide, in the therapeutic concentration, enhances GABA-activated currents in cultured neurons of rat cerebral cortex. Epilepsy Res. 1994 Oct;19(2):89-97. [7843172 ]