| Record Information |
|---|
| Version | 2.0 |
|---|
| Creation Date | 2009-06-22 16:08:41 UTC |
|---|
| Update Date | 2014-12-24 20:24:43 UTC |
|---|
| Accession Number | T3D1838 |
|---|
| Identification |
|---|
| Common Name | Silver selenite |
|---|
| Class | Small Molecule |
|---|
| Description | Silver selenite is a chemical compound of silver and selenium. Selenium is a nonmetal element with the atomic number 34 and the chemical symbol Se. Selenium rarely occurs in its elemental state in nature and is usually found in sulfide ores such as pyrite, partially replacing the sulfur in the ore matrix. It may also be found in silver, copper, lead, and nickel minerals. Though selenium salts are toxic in large amounts, trace amounts of the element are necessary for cellular function in most animals, forming the active center of the enzymes glutathione peroxidase, thioredoxin reductase, and three known deiodinase enzymes. Silver is a metallic element with the chemical symbol Ag and atomic number 47. It occurs naturally in its pure, free form, as an alloy with gold and other metals, and in minerals such as argentite and chlorargyrite. (9, 10, 8) |
|---|
| Compound Type | - Industrial/Workplace Toxin
- Inorganic Compound
- Pollutant
- Silver Compound
- Synthetic Compound
|
|---|
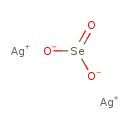
| Chemical Structure | |
|---|
| Synonyms | | Synonym | | Disilver selenite | | Silver(I) selenite |
|
|---|
| Chemical Formula | Ag2O3Se |
|---|
| Average Molecular Mass | 342.690 g/mol |
|---|
| Monoisotopic Mass | 341.711 g/mol |
|---|
| CAS Registry Number | 7784-05-6 |
|---|
| IUPAC Name | disilver(1+) ion selenite |
|---|
| Traditional Name | disilver(1+) ion selenite |
|---|
| SMILES | [Ag+].[Ag+].[O-][Se]([O-])=O |
|---|
| InChI Identifier | InChI=1S/2Ag.H2O3Se/c;;1-4(2)3/h;;(H2,1,2,3)/q2*+1;/p-2 |
|---|
| InChI Key | InChIKey=WQIJNCUKAOHNPM-UHFFFAOYSA-L |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of inorganic compounds known as transition metal selenites. These are inorganic compounds in which the largest oxoanion is selenite, and in which the heaviest atom not in an oxoanion is a transition metal. |
|---|
| Kingdom | Inorganic compounds |
|---|
| Super Class | Mixed metal/non-metal compounds |
|---|
| Class | Transition metal oxoanionic compounds |
|---|
| Sub Class | Transition metal selenites |
|---|
| Direct Parent | Transition metal selenites |
|---|
| Alternative Parents | |
|---|
| Substituents | - Transition metal selenite
- Inorganic silver salt
- Inorganic oxide
- Inorganic salt
|
|---|
| Molecular Framework | Not Available |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Exogenous |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Appearance | White powder. |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available | | LogP | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | Deposition Date | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0009000000-9ed8dae940f19aa5ceb8 | 2016-06-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0006-0009000000-9ed8dae940f19aa5ceb8 | 2016-06-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0006-0009000000-9ed8dae940f19aa5ceb8 | 2016-06-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-0009000000-ea327191e38a077d840f | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0006-0009000000-ea327191e38a077d840f | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-0009000000-ea327191e38a077d840f | 2016-08-03 | View Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Oral (7) ; inhalation (7) ; dermal (7) |
|---|
| Mechanism of Toxicity | Selenium readily substitutes for sulfur in biomolecules and in many biochemical reactions, especially when the concentration of selenium is high and the concentration of sulfur is low. Inactivation of the sulfhydryl enzymes necessary for oxidative reactions in cellular respiration, through effects on mitochondrial and microsomal electron transport, might contribute to acute selenium toxicity. Selenomethionine (a common organic selenium compound) also appears to randomly substitute for methionine in protein synthesis. This substitution may affect the structure and functionability of the protein, for example, by altering disulfide bridges. Inorganic forms of selenium appear to react with tissue thiols by redox catalysis, resulting in formation of reactive oxygen species and causing damage by oxidative stress. Metallic silver is oxidized and may deposit in the tissues, causing arygria. The silver ion is known to inhibit glutathione peroxidase and NA+,K+-ATPase activity, disrupting selenium-catalyzed sulfhydryl oxidation-reduction reactions and intracellular ion concentrations, respectively. Silver nanoparticles are believed to disrupt the mitochondrial respiratory chain, causing oxidative stress, reduced ATP synthesis, and DNA damage. (9, 1, 2, 3, 4, 7) |
|---|
| Metabolism | Selenium may be absorbed through inhalation and ingestion, while some selenium compounds may also be absorbed dermally. Once in the body, selenium is distributed mainly to the liver and kidney. Selenium is an essential micronutrient and is a component of glutathione peroxidase, iodothyronine 5'-deiodinases, and thioredoxin reductase. Organic selenium is first metabolized into inorganic selenium. Inorganic selenium is reduced stepwise to the intermediate hydrogen selenide, which is either incorporated into selenoproteins after being transformed to selenophosphate and selenocysteinyl tRNA or excreted into the urine after being transformed into methylated metabolites of selenide. Elemental selenium is also methylated before excretion. Selenium is primarily eliminated in the urine and feces, but certain selenium compounds may also be exhaled. Silver compounds can also be absorbed orally and dermally. It distributes throughout the body in the blood, particularily to the liver. Insoluble silver salts are transformed into soluble silver sulfide albuminates, bind to amino or carboxyl groups in RNA, DNA, and proteins, or are reduced to metallic silver by ascorbic acid or catecholamines. Metallic silver is oxidized and may deposit in the tissues, causing arygria. Silver is eliminated primarily in the faeces. (9, 7) |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | 3, not classifiable as to its carcinogenicity to humans. (6) |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Chronic Oral: 0.005 mg/kg/day (5) |
|---|
| Health Effects | Chronic oral exposure to high concentrations of selenium compounds can produce a disease called selenosis. The major signs of selenosis are hair loss, nail brittleness, and neurological abnormalities (such as numbness and other odd sensations in the extremities). Animal studies have shown that selenium may also affect sperm production and the female reproductive cycle. Exposure to high levels of silver for a long period of time may result in a condition called arygria, a blue-gray discoloration of the skin and other body tissues. Argyria is a permanent effect but does not appear to be harmful to health. While silver itself is not toxic, most silver salts are, and may damage the liver, kidney, and central nervous system, as well as be carcinogenic. (9, 10, 11, 7) |
|---|
| Symptoms | Short-term oral exposure to high concentrations of selenium may cause nausea, vomiting, and diarrhea. Brief exposures to high levels of elemental selenium or selenium dioxide in air can result in respiratory tract irritation, bronchitis, difficulty breathing, and stomach pains. Longer-term exposure to either of these air-borne forms can cause respiratory irritation, bronchial spasms, and coughing. Exposure to high levels of silver for a long period of time may result in a condition called arygria, a blue-gray discoloration of the skin and other body tissues. Argyria is a permanent effect but does not appear to be harmful to health. Exposure to high levels of silver in the air has resulted in breathing problems, lung and throat irritation, and stomach pains. Skin contact with silver can cause mild allergic reactions such as rash, swelling, and inflammation in some people. (9, 7) |
|---|
| Treatment | Not Available |
|---|
| Normal Concentrations |
|---|
| Not Available |
|---|
| Abnormal Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| PubChem Compound ID | 15520661 |
|---|
| ChEMBL ID | Not Available |
|---|
| ChemSpider ID | 14668373 |
|---|
| KEGG ID | Not Available |
|---|
| UniProt ID | Not Available |
|---|
| OMIM ID | |
|---|
| ChEBI ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| CTD ID | Not Available |
|---|
| Stitch ID | Silver selenite |
|---|
| PDB ID | Not Available |
|---|
| ACToR ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | - Bianchini A, Playle RC, Wood CM, Walsh PJ: Mechanism of acute silver toxicity in marine invertebrates. Aquat Toxicol. 2005 Mar 25;72(1-2):67-82. Epub 2004 Dec 29. [15748748 ]
- AshaRani PV, Low Kah Mun G, Hande MP, Valiyaveettil S: Cytotoxicity and genotoxicity of silver nanoparticles in human cells. ACS Nano. 2009 Feb 24;3(2):279-90. doi: 10.1021/nn800596w. [19236062 ]
- Kim S, Choi JE, Choi J, Chung KH, Park K, Yi J, Ryu DY: Oxidative stress-dependent toxicity of silver nanoparticles in human hepatoma cells. Toxicol In Vitro. 2009 Sep;23(6):1076-84. doi: 10.1016/j.tiv.2009.06.001. Epub 2009 Jun 7. [19508889 ]
- Dillard CJ, Tappel AL: Mercury, silver, and gold inhibition of selenium-accelerated cysteine oxidation. J Inorg Biochem. 1986 Sep;28(1):13-20. [3760861 ]
- ATSDR - Agency for Toxic Substances and Disease Registry (2001). Minimal Risk Levels (MRLs) for Hazardous Substances. U.S. Public Health Service in collaboration with U.S. Environmental Protection Agency (EPA). [Link]
- International Agency for Research on Cancer (2014). IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. [Link]
- ATSDR - Agency for Toxic Substances and Disease Registry (2003). Toxicological profile for selenium. U.S. Public Health Service in collaboration with U.S. Environmental Protection Agency (EPA). [Link]
- Wikipedia. Selenium. Last Updated 7 June 2009. [Link]
- ATSDR - Agency for Toxic Substances and Disease Registry (1990). Toxicological profile for silver. U.S. Public Health Service in collaboration with U.S. Environmental Protection Agency (EPA). [Link]
- Wikipedia. Silver. Last updated Dec 2014. [Link]
- International Programme on Chemical Safety (IPCS) INCHEM (1977). WHO Food Additive Series No. 12: Silver. [Link]
|
|---|
| Gene Regulation |
|---|
| Up-Regulated Genes | Not Available |
|---|
| Down-Regulated Genes | Not Available |
|---|