| Record Information |
|---|
| Version | 2.0 |
|---|
| Creation Date | 2009-06-24 15:20:02 UTC |
|---|
| Update Date | 2014-12-24 20:24:52 UTC |
|---|
| Accession Number | T3D1910 |
|---|
| Identification |
|---|
| Common Name | 2,3-Dichloropropene |
|---|
| Class | Small Molecule |
|---|
| Description | 2,3-dichloropropene is a straw-colored liquid form of dichloropropene (6). |
|---|
| Compound Type | - Chloropropene
- Food Toxin
- Lachrymator
- Organic Compound
- Organochloride
- Pesticide
- Synthetic Compound
|
|---|
| Chemical Structure | |
|---|
| Synonyms | | Synonym | | 1,2-Dichloro-2-propene | | 2,3-Dichloro-1-propene | | 2,3-dichloroprop-1-ene | | 2,3-Dichloropropene-(1) | | 2,3-Dichloropropylene | | 2-Chloroallyl chloride | | 3-Dichloro-propene | | Propylene, 2,3-dichloro |
|
|---|
| Chemical Formula | C3H4Cl2 |
|---|
| Average Molecular Mass | 110.970 g/mol |
|---|
| Monoisotopic Mass | 109.969 g/mol |
|---|
| CAS Registry Number | 78-88-6 |
|---|
| IUPAC Name | 2,3-dichloroprop-1-ene |
|---|
| Traditional Name | 2,3-dichloropropene |
|---|
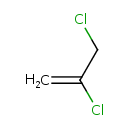
| SMILES | ClCC(Cl)=C |
|---|
| InChI Identifier | InChI=1S/C3H4Cl2/c1-3(5)2-4/h1-2H2 |
|---|
| InChI Key | InChIKey=FALCMQXTWHPRIH-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as vinyl chlorides. These are vinyl halides in which a chlorine atom is bonded to an sp2-hybridised carbon atom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organohalogen compounds |
|---|
| Class | Vinyl halides |
|---|
| Sub Class | Vinyl chlorides |
|---|
| Direct Parent | Vinyl chlorides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Chloroalkene
- Haloalkene
- Vinyl chloride
- Hydrocarbon derivative
- Organochloride
- Alkyl halide
- Alkyl chloride
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Exogenous |
|---|
| Cellular Locations | |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Liquid |
|---|
| Appearance | Straw-colored liquid (6). |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | 10°C | | Boiling Point | Not Available | | Solubility | 2.15 mg/mL at 25°C [MACKAY,D & SHIU,WY (1981)] | | LogP | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | Deposition Date | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-0900000000-eb2d73fbb8c162bd6f17 | 2016-08-02 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03di-0900000000-e72857adbeb1f13bb9a5 | 2016-08-02 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-004l-9000000000-41bc9baf5ac4bb15d3ef | 2016-08-02 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-0900000000-e2678442c752a1d457a3 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-1900000000-0cfd092dc3b04e67654f | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-05fr-9500000000-717061a020942a8e2428 | 2016-08-03 | View Spectrum | | MS | Mass Spectrum (Electron Ionization) | splash10-004r-9100000000-6ac4dac13cef5369d270 | 2014-09-20 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 90 MHz, CDCl3, experimental) | Not Available | 2014-09-20 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 25.16 MHz, CDCl3, experimental) | Not Available | 2014-09-23 | View Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Inhalation (6) ; oral (6) ; dermal (6) ; eye contact (6). |
|---|
| Mechanism of Toxicity | The primary toxic effects of 2,3-dichloropropene are portal-of-entry effects resulting from the chemical reactivity of the compound and its physicochemical properties. Repeated irritation results in a hyperplastic response in the target tissues. It is likely that depletion of glutathione would block the major detoxification pathway for 2,3-dichloropropene, resulting in increased toxicity of organs such as the liver and kidney because of binding of reactive intermediates to macromolecules in cells (6). |
|---|
| Metabolism | The major pathway is a detoxifying conjugation to glutathione, leading to the elimination of mercapturic
acid metabolites in the urine. Two secondary pathways result in the formation of mutagenic metabolites. One involves cytochrome P450-induced formation of an epoxide that undergoes spontaneous rearrangement to form the mutagen 1,3-dichloroacetone. The other involves hydrolysis and dechlorination to form an intermediate
(2-chloroallyl alcohol) that can either be detoxified by conjugation to glucuronic acid or bioactivated by
alcohol dehydrogenase to form the mutagen 2-chloroacrolein. It is evident that depletion of glutathione stores, more likely to occur under bolus exposure conditions, would result in the formation of proportionally more mutagenic metabolites (6). |
|---|
| Toxicity Values | LD50: 285 mg/kg (Oral, Rat) (6)
LD50: 1913 mg/kg (Dermal, Rat) (6) |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | No indication of carcinogenicity (not listed by IARC). (5) |
|---|
| Uses/Sources | Breathing in contaminated air; drinking contaminated water; eating contaminated food; dermal and eye exposure (6). |
|---|
| Minimum Risk Level | Acute Inhalation: 0.002 ppm (Rabbit) (6) |
|---|
| Health Effects | Ingestion of 2,3-D can lead to developed gastrointestinal distress, adult respiratory distress syndrome, hematological and hepatorenal functional impairment, acute gastrointestinal distress with pulmonary congestion and edema, central nervous depression, perhaps even in the absence of impaired oxygen uptake. Moreover, this can lead to death. Coma may occur rapidly after inhalation. Severe skin irritation with marked inflammatory response of epidermis can underlying tissues can follow dermal exposure. By any route, possible late injuries to liver, kidneys and heart (3). |
|---|
| Symptoms | Symptoms occuring after inhalation include gasping, refusal to breathe, coughing, substernal pain; lacrimation and headache are prominant. After inhalation exposures, malaise, headache, chest and abdominal discomfort and irritability can persist during weeks or years. Moreover, Irritation of eyes and upper respiratory mucosa appears promptly after exposure to concentrated vapors. Ingestion can cause cough, sore throat, headache, dizziness, nausea, vomiting, unconsciousness, and laboured breathing (3, 6). |
|---|
| Treatment | Following oral exposure, administer charcoal as a slurry. Monitor liver and kidney function; elevations may not be seen for several days. Following eye exposure, irrigate exposed eyes with copious amounts of room temperature water for at least 15 minutes. If irritation, pain, swelling, lacrimation, or photophobia persist, the patient should be seen in a health care facility. Following dermal exposure, remove contaminated clothing and wash exposed area thoroughly with soap and water. Following inhalation, move patient to fresh air, even though initial symptoms and signs are mild; keep the victim quiet, in a semi-reclining position. Minimum physical activity limits the likehood of pulmonary edema. If victim is not breathing, clear the airway of secretions and resuscitate with positive pressure oxygen apparatus. If this is not available, use chest compression to sustain respiration. (2, 1) |
|---|
| Normal Concentrations |
|---|
| Not Available |
|---|
| Abnormal Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| PubChem Compound ID | 6565 |
|---|
| ChEMBL ID | CHEMBL156075 |
|---|
| ChemSpider ID | 6317 |
|---|
| KEGG ID | Not Available |
|---|
| UniProt ID | Not Available |
|---|
| OMIM ID | |
|---|
| ChEBI ID | Not Available |
|---|
| BioCyc ID | CPD-9112 |
|---|
| CTD ID | C043065 |
|---|
| Stitch ID | 2,3-Dichloropropene |
|---|
| PDB ID | Not Available |
|---|
| ACToR ID | 6801 |
|---|
| Wikipedia Link | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | T3D1910.pdf |
|---|
| General References | - Dalfo D, Marques N, Albalat R: Analysis of the NADH-dependent retinaldehyde reductase activity of amphioxus retinol dehydrogenase enzymes enhances our understanding of the evolution of the retinol dehydrogenase family. FEBS J. 2007 Jul;274(14):3739-52. Epub 2007 Jul 2. [17608724 ]
- Rumack BH (2009). POISINDEX(R) Information System. Englewood, CO: Micromedex, Inc. CCIS Volume 141, edition expires Aug, 2009.
- Gosselin RE, Smith RP, and Hodge HC (1984). Clinical Toxicology of Commercial Products. 5th ed. Baltimore: Williams and Wilkins.
- Sullivan JB Jr. and Krieger GR (eds) (1999). Hazardous Materials Toxicology-Clinical Principles of Environmental Health. Baltimore, MD: Williams and Wilkins.
- International Agency for Research on Cancer (2014). IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. [Link]
- ATSDR - Agency for Toxic Substances and Disease Registry (2008). Toxicological profile for dichloropropenes. U.S. Public Health Service in collaboration with U.S. Environmental Protection Agency (EPA). [Link]
|
|---|
| Gene Regulation |
|---|
| Up-Regulated Genes | Not Available |
|---|
| Down-Regulated Genes | Not Available |
|---|