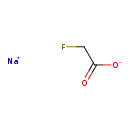| Record Information |
|---|
| Version | 2.0 |
|---|
| Creation Date | 2009-08-12 14:05:03 UTC |
|---|
| Update Date | 2014-12-24 20:26:10 UTC |
|---|
| Accession Number | T3D3580 |
|---|
| Identification |
|---|
| Common Name | Sodium fluoroacetate |
|---|
| Class | Small Molecule |
|---|
| Description | Sodium fluoroacetate is an organofluorine compound and a derivative of fluoroacetic acid. Sodium fluoroacetate is known under its brand name "1080". Sodium fluoroacetate occurs naturally as an anti-herbivore metabolite in various plants but can also be produced synthetically. Fluoroacetate is highly toxic to mammals and insects; it is used as a pesticide especially for mammalian pest species. The more common fluorinated acetic acid, trifluoroacetic acid and its sodium salt are far less toxic. (2) |
|---|
| Compound Type | - Organic Compound
- Organofluoride
- Pesticide
- Synthetic Compound
|
|---|
| Chemical Structure | |
|---|
| Synonyms | | Synonym | | Fluoroacetic acid sodium salt | | Natriumfluoracetat | | Sodium fluoroacetic acid | | Sodium monofluoroacetate |
|
|---|
| Chemical Formula | C2H2FNaO2 |
|---|
| Average Molecular Mass | 100.024 g/mol |
|---|
| Monoisotopic Mass | 99.994 g/mol |
|---|
| CAS Registry Number | 62-74-8 |
|---|
| IUPAC Name | sodium 2-fluoroacetate |
|---|
| Traditional Name | sodium fluoroacetate |
|---|
| SMILES | [Na+].[O-]C(=O)CF |
|---|
| InChI Identifier | InChI=1S/C2H3FO2.Na/c3-1-2(4)5;/h1H2,(H,4,5);/q;+1/p-1 |
|---|
| InChI Key | InChIKey=JGFYQVQAXANWJU-UHFFFAOYSA-M |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as alpha-halocarboxylic acids. These are carboxylic acids containing a halogen atom bonded to the alpha carbon atom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Alpha-halocarboxylic acids and derivatives |
|---|
| Direct Parent | Alpha-halocarboxylic acids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Alpha-halocarboxylic acid
- Carboxylic acid salt
- Organic metal halide
- Carboxylic acid
- Organic alkali metal salt
- Monocarboxylic acid or derivatives
- Alkyl fluoride
- Organic salt
- Organooxygen compound
- Organofluoride
- Organohalogen compound
- Organic sodium salt
- Hydrocarbon derivative
- Organic oxide
- Carbonyl group
- Organic oxygen compound
- Alkyl halide
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Exogenous |
|---|
| Cellular Locations | |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Appearance | Odourless white powder. (1) |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | 200°C (325.15°K) | | Boiling Point | Not Available | | Solubility | Not Available | | LogP | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | Deposition Date | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-6900000000-569a907c8575fb953c9d | 2019-02-23 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0pb9-9500000000-f6612ed8e54151211376 | 2019-02-23 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0006-9000000000-c38f4fc2aa6d983aaf82 | 2019-02-23 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-9000000000-b1353035c7679a46035c | 2019-02-23 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0002-9000000000-ec2e810004e52259cc30 | 2019-02-23 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0002-9000000000-69a83ef6cd4d50dd12c6 | 2019-02-23 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, experimental) | Not Available | 2014-09-20 | View Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Fluoroacetate is similar to acetate, which has a pivotal role in cellular metabolism. Fluoroacetate disrupts the citric acid cycle by combining with coenzyme A to form fluoroacetyl CoA. Fluoroacetyl CoA then reacts with citrate synthase to produce fluorocitrate. A metabolite of fluorocitrate binds very tightly to aconitase, thereby halting the citric acid cycle. This inhibition results in an accumulation of citrate in the blood which deprives cells of energy. (2) |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | No indication of carcinogenicity to humans (not listed by IARC). |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | The symptoms of sodium fluoroacetate poisoning normally appear between 30 minutes and three hours after exposure. Initial symptoms typically include nausea, vomiting and abdominal pain; sweating, confusion and agitation follow. In significant poisoning cardiac abnormalities including tachycardia or bradycardia, hypotension and ECG changes develop. Neurological effects include muscle twitching and seizures. Consciousness becomes progressively impaired after a few hours leading to coma. Death is generally due to ventricular arrhythmias, progressive hypotension unresponsive to treatment, and secondary lung infections. Sub-lethal doses of sodium fluoroacetate may cause damage to tissues with high energy needs, in particular, the brain, gonads, heart, lungs and fetus. Sub-lethal doses are typically completely metabolised and excreted within four days. (2) |
|---|
| Treatment | Because of the biochemical interference in the citric acid cycle, sodium fluoroacetate poisoning is very difficult to treat. Once clinical symptoms are exhibited, the citric acid cycle has shut down. Effective antidotes are unknown. Research in animals has shown that the use of glyceryl monoacetate can prevent problems if given after ingestion of sodium fluoroacetate. In clinical cases, use of muscle relaxants, anti-convulsants, mechanical ventilation, and other supportive measures may all be required. Few animals or people have been treated successfully after significant sodium fluoroacetate ingestions. (2) |
|---|
| Normal Concentrations |
|---|
| Not Available |
|---|
| Abnormal Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| PubChem Compound ID | 16212360 |
|---|
| ChEMBL ID | CHEMBL369611 |
|---|
| ChemSpider ID | 5893 |
|---|
| KEGG ID | C18588 |
|---|
| UniProt ID | Not Available |
|---|
| OMIM ID | |
|---|
| ChEBI ID | 38699 |
|---|
| BioCyc ID | Not Available |
|---|
| CTD ID | Not Available |
|---|
| Stitch ID | Sodium fluoroacetate |
|---|
| PDB ID | Not Available |
|---|
| ACToR ID | 6534 |
|---|
| Wikipedia Link | Sodium_fluoroacetate |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | T3D3580.pdf |
|---|
| General References | - HSDB: Hazardous Substances Data Bank. National Library of Medicine (2001). [Link]
- Wikipedia. Sodium fluoroacetate. Last Updated 8 August 2009. [Link]
|
|---|
| Gene Regulation |
|---|
| Up-Regulated Genes | Not Available |
|---|
| Down-Regulated Genes | Not Available |
|---|