| Record Information |
|---|
| Version | 2.0 |
|---|
| Creation Date | 2014-10-14 21:18:31 UTC |
|---|
| Update Date | 2014-12-24 20:27:01 UTC |
|---|
| Accession Number | T3D4980 |
|---|
| Identification |
|---|
| Common Name | Atorvastatin |
|---|
| Class | Small Molecule |
|---|
| Description | Atorvastatin (Lipitor) is a member of the drug class known as statins. It is used for lowering cholesterol. Atorvastatin is a competitive inhibitor of hydroxymethylglutaryl-coenzyme A (HMG-CoA) reductase, the rate-determining enzyme in cholesterol biosynthesis via the mevalonate pathway. HMG-CoA reductase catalyzes the conversion of HMG-CoA to mevalonate. Atorvastatin acts primarily in the liver. Decreased hepatic cholesterol levels increases hepatic uptake of cholesterol and reduces plasma cholesterol levels. |
|---|
| Compound Type | - Anticholesteremic Agent
- Drug
- Hydroxymethylglutaryl-CoA Reductase Inhibitor
- Metabolite
- Synthetic Compound
|
|---|
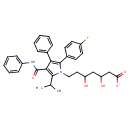
| Chemical Structure | |
|---|
| Synonyms | | Synonym | | Atogal | | Atorvastatin acid | | Atorvastatin calcium | | Cardyl | | Faboxim | | Hipolixan | | Lipitor | | Lipotropic | | Lipovastatinklonal | | Liprimar | | Lowden | | Normalip | | Sincol | | Sortis | | Torvacard | | Torvast | | Totalip | | Tulip | | Vastina | | Xanator | | Xarator | | Zurinel |
|
|---|
| Chemical Formula | C33H34FN2O5 |
|---|
| Average Molecular Mass | 557.632 g/mol |
|---|
| Monoisotopic Mass | 557.246 g/mol |
|---|
| CAS Registry Number | 134523-00-5 |
|---|
| IUPAC Name | 7-[2-(4-fluorophenyl)-3-phenyl-4-(phenylcarbamoyl)-5-(propan-2-yl)-1H-pyrrol-1-yl]-3,5-dihydroxyheptanoate |
|---|
| Traditional Name | 7-[2-(4-fluorophenyl)-5-isopropyl-3-phenyl-4-(phenylcarbamoyl)pyrrol-1-yl]-3,5-dihydroxyheptanoate |
|---|
| SMILES | CC(C)C1=C(C(O)=NC2=CC=CC=C2)C(=C(N1CCC(O)CC(O)CC([O-])=O)C1=CC=C(F)C=C1)C1=CC=CC=C1 |
|---|
| InChI Identifier | InChI=1/C33H35FN2O5/c1-21(2)31-30(33(41)35-25-11-7-4-8-12-25)29(22-9-5-3-6-10-22)32(23-13-15-24(34)16-14-23)36(31)18-17-26(37)19-27(38)20-28(39)40/h3-16,21,26-27,37-38H,17-20H2,1-2H3,(H,35,41)(H,39,40)/p-1 |
|---|
| InChI Key | InChIKey=XUKUURHRXDUEBC-UHFFFAOYNA-M |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as aminophenyl ethers. These are aromatic compounds that contain a phenol ether, which carries an amine group on the benzene ring. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Phenol ethers |
|---|
| Sub Class | Aminophenyl ethers |
|---|
| Direct Parent | Aminophenyl ethers |
|---|
| Alternative Parents | |
|---|
| Substituents | - Aminophenyl ether
- Methoxyaniline
- Phenoxy compound
- Anisole
- Aniline or substituted anilines
- Methoxybenzene
- Alkyl aryl ether
- Monocyclic benzene moiety
- Ether
- Organopnictogen compound
- Primary amine
- Organooxygen compound
- Organonitrogen compound
- Organic nitrogen compound
- Organic oxygen compound
- Amine
- Hydrocarbon derivative
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Endogenous |
|---|
| Cellular Locations | |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | 159.2-160.7 °C | | Boiling Point | Not Available | | Solubility | Sodium salt soluble in water, 20.4 ug/mL (pH 2.1), 1.23 mg/mL (pH 6.0) | | LogP | 5.7 |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | Deposition Date | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0aor-0000090000-401e9c7a2f170820f729 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014j-0000090000-647b0fa490907c33201c | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-000j-4200790000-a372fe1f0093d1e98cd3 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-2100090000-fe1fdeff5a26f7f83882 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-8400090000-7dd2f7f373fb6fd5de88 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-9500120000-34427c424cc44ee99074 | 2016-08-03 | View Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Atorvastatin is rapidly absorbed after oral administration with maximum plasma concentrations achieved in 1 to 2 hours. The absolute bioavailability of atorvastatin (parent drug) is approximately 14% and the systemic availability of HMG-CoA reductase inhibitory activity is approximately 30%. The low systemic bioavailability is due to presystemic clearance by gastrointestinal mucosa and first-pass metabolism in the liver. |
|---|
| Mechanism of Toxicity | Atorvastatin selectively and competitively inhibits the hepatic enzyme HMG-CoA reductase. As HMG-CoA reductase is responsible for converting HMG-CoA to mevalonate in the cholesterol biosynthesis pathway, this results in a subsequent decrease in hepatic cholesterol levels. Decreased hepatic cholesterol levels stimulates upregulation of hepatic LDL-C receptors which increases hepatic uptake of LDL-C and reduces serum LDL-C concentrations. |
|---|
| Metabolism | Atorvastatin is extensively metabolized to ortho- and parahydroxylated derivatives and various beta-oxidation products. In vitro inhibition of HMG-CoA reductase by ortho- and parahydroxylated metabolites is equivalent to that of atorvastatin. Approximately 70% of circulating inhibitory activity for HMG-CoA reductase is attributed to active metabolites. CYP3A4 is also involved in the metabolism of atorvastatin. |
|---|
| Toxicity Values | Generally well-tolerated. Side effects may include myalgia, constipation, asthenia, abdominal pain, and nausea. Other possible side effects include myotoxicity (myopathy, myositis, rhabdomyolysis) and hepatotoxicity. To avoid toxicity in Asian patients, lower doses should be considered. |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | No indication of carcinogenicity to humans (not listed by IARC). |
|---|
| Uses/Sources | May be used as primary prevention in individuals with multiple risk factors for coronary heart disease (CHD) and as secondary prevention in individuals with CHD to reduce the risk of myocardial infarction (MI), stroke, angina, and revascularization procedures. May be used to reduce the risk of cardiovascular events in patients with acute coronary syndrome (ACS). May be used in the treatment of primary hypercholesterolemia and mixed dyslipidemia, homozygous familial hypercholesterolemia, primary dysbetalipoproteinemia, and/or hypertriglyeridemia as an adjunct to dietary therapy to decrease serum total and low-density lipoprotein cholesterol (LDL-C), apolipoprotein B (apoB), and triglyceride concentrations, while increasing high-density lipoprotein cholesterol (HDL-C) levels. |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Normal Concentrations |
|---|
| Not Available |
|---|
| Abnormal Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB01076 |
|---|
| HMDB ID | HMDB05006 |
|---|
| PubChem Compound ID | 60823 |
|---|
| ChEMBL ID | CHEMBL1487 |
|---|
| ChemSpider ID | 54810 |
|---|
| KEGG ID | C06834 |
|---|
| UniProt ID | Not Available |
|---|
| OMIM ID | |
|---|
| ChEBI ID | 2910 |
|---|
| BioCyc ID | Not Available |
|---|
| CTD ID | Not Available |
|---|
| Stitch ID | Not Available |
|---|
| PDB ID | 117 |
|---|
| ACToR ID | Not Available |
|---|
| Wikipedia Link | Atorvastatin |
|---|
| References |
|---|
| Synthesis Reference | Zlatko Pflaum, “Process for the preparation of amorphous atorvastatin.” U.S. Patent US20020183527, issued December 05, 2002. |
|---|
| MSDS | T3D4980.pdf |
|---|
| General References | - Rouleau J: Improved outcome after acute coronary syndromes with an intensive versus standard lipid-lowering regimen: results from the Pravastatin or Atorvastatin Evaluation and Infection Therapy-Thrombolysis in Myocardial Infarction 22 (PROVE IT-TIMI 22) trial. Am J Med. 2005 Dec;118 Suppl 12A:28-35. [16356805 ]
- Maggon K: Best-selling human medicines 2002-2004. Drug Discov Today. 2005 Jun 1;10(11):739-42. [15922927 ]
- Taylor PJ, Kubler PA, Lynch SV, Allen J, Butler M, Pillans PI: Effect of atorvastatin on cyclosporine pharmacokinetics in liver transplant recipients. Ann Pharmacother. 2004 Feb;38(2):205-8. Epub 2003 Dec 19. [14742751 ]
- Funatsu T, Suzuki K, Goto M, Arai Y, Kakuta H, Tanaka H, Yasuda S, Ida M, Nishijima S, Miyata K: Prolonged inhibition of cholesterol synthesis by atorvastatin inhibits apo B-100 and triglyceride secretion from HepG2 cells. Atherosclerosis. 2001 Jul;157(1):107-15. [11427209 ]
- Tannous M, Cheung R, Vignini A, Mutus B: Atorvastatin increases ecNOS levels in human platelets of hyperlipidemic subjects. Thromb Haemost. 1999 Nov;82(5):1390-4. [10595624 ]
- Naoumova RP, Dunn S, Rallidis L, Abu-Muhana O, Neuwirth C, Rendell NB, Taylor GW, Thompson GR: Prolonged inhibition of cholesterol synthesis explains the efficacy of atorvastatin. J Lipid Res. 1997 Jul;38(7):1496-500. [9254075 ]
- Izar MC: [Hypolipidemic treatment under special conditions: posttransplant and/or immunosuppressive therapy]. Arq Bras Cardiol. 2005 Oct;85 Suppl 5:50-7. Epub 2006 Jan 2. [16400400 ]
- Hwang YS, Tsai WC, Lu YH, Lin CC, Chen YF: Effect of atorvastatin on the expression of CD40 ligand and P-selectin on platelets in patients with hypercholesterolemia. Am J Cardiol. 2004 Aug 1;94(3):364-6. [15276107 ]
- Bruni F, Pasqui AL, Pastorelli M, Bova G, Cercignani M, Palazzuoli A, Sawamura T, Gioffre WR, Auteri A, Puccetti L: Different effect of statins on platelet oxidized-LDL receptor (CD36 and LOX-1) expression in hypercholesterolemic subjects. Clin Appl Thromb Hemost. 2005 Oct;11(4):417-28. [16244767 ]
- Fenton JW 2nd, Brezniak DV, Ofosu FA, Shen GX, Jacobson JR, Garcia JG: Statins and thrombin. Curr Drug Targets Cardiovasc Haematol Disord. 2005 Apr;5(2):115-20. [15853752 ]
- Pokrovskaia EV, Vaulin NA, Gratsianskii NA, Averkov OV, Deev AD: [Markers of inflammation and platelet aggregation in patients with non ST elevation acute coronary syndrome treated with atorvastatin or pravastatin]. Kardiologiia. 2003;43(1):7-18. [12891281 ]
- Funatsu T, Kakuta H, Tanaka H, Arai Y, Suzuki K, Miyata K: [Atorvastatin (Lipitor): a review of its pharmacological and clinical profile]. Nihon Yakurigaku Zasshi. 2001 Jan;117(1):65-76. [11233299 ]
- Tousoulis D, Bosinakou E, Kotsopoulou M, Antoniades C, Katsi V, Stefanadis C: Effects of early administration of atorvastatin treatment on thrombotic process in normocholesterolemic patients with unstable angina. Int J Cardiol. 2006 Jan 26;106(3):333-7. [16337041 ]
- Malinowski JM: Atorvastatin: a hydroxymethylglutaryl-coenzyme A reductase inhibitor. Am J Health Syst Pharm. 1998 Nov 1;55(21):2253-67; quiz 2302-3. [9825877 ]
- DailyMed FDA Label [Link]
|
|---|
| Gene Regulation |
|---|
| Up-Regulated Genes | Not Available |
|---|
| Down-Regulated Genes | Not Available |
|---|