| Record Information |
|---|
| Version | 2.0 |
|---|
| Creation Date | 2009-07-03 22:03:17 UTC |
|---|
| Update Date | 2014-12-24 20:25:34 UTC |
|---|
| Accession Number | T3D2478 |
|---|
| Identification |
|---|
| Common Name | Levofloxacin |
|---|
| Class | Small Molecule |
|---|
| Description | Levofloxacin is a synthetic fluoroquinolone antibacterial agent that inhibits the supercoiling activity of bacterial DNA gyrase, halting DNA replication. Levofloxacin is marketed by Ortho-McNeil under the trade name Levaquin. Chemically, levofloxacin is the S-enantiomer (L-isomer) of ofloxacin. |
|---|
| Compound Type | - Amide
- Amine
- Anti-Bacterial Agent
- Anti-Infective Agent, Urinary
- Drug
- Ester
- Ether
- Food Toxin
- Metabolite
- Nucleic Acid Synthesis Inhibitor
- Organic Compound
- Organofluoride
- Quinolone
- Synthetic Compound
|
|---|
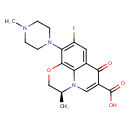
| Chemical Structure | |
|---|
| Synonyms | | Synonym | | (-)-Ofloxacin | | (3S)-(-)-9-Fluoro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-2,3-dihydro-7H-pyrido[1,2,3-de][1,4]benzoxazine-6-carboxylic acid | | (S)-(-)-9-Fluoro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-2,3-dihydro-7H-pyrido[1,2,3-de][1,4]benzooxazine-6-carboxylic acid | | (S)-9-Fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-7H-pyrido(1,2,3-de)-1,4-benzoxazine-6-carboxylic acid | | (S)-Ofloxacin | | Cravit | | Elequine | | Floxel | | Floxin | | Iquix | | L-ofloxacin | | Leroxacin | | Levaquin | | Levofloxacin hydrate | | Levofloxacine | | Levofloxacino | | Levofloxacinum | | Levokacin | | Levox | | Levoxacin | | Mosardal | | Nofaxin | | Ofloxacin | | Ofloxacin S-(-)-form | | Quixin | | Reskuin | | Tavanic |
|
|---|
| Chemical Formula | C18H20FN3O4 |
|---|
| Average Molecular Mass | 361.368 g/mol |
|---|
| Monoisotopic Mass | 361.144 g/mol |
|---|
| CAS Registry Number | 100986-85-4 |
|---|
| IUPAC Name | (2S)-7-fluoro-2-methyl-6-(4-methylpiperazin-1-yl)-10-oxo-4-oxa-1-azatricyclo[7.3.1.0^{5,13}]trideca-5(13),6,8,11-tetraene-11-carboxylic acid |
|---|
| Traditional Name | (2S)-7-fluoro-2-methyl-6-(4-methylpiperazin-1-yl)-10-oxo-4-oxa-1-azatricyclo[7.3.1.0^{5,13}]trideca-5(13),6,8,11-tetraene-11-carboxylic acid |
|---|
| SMILES | [H][C@]1(C)COC2=C3N1C=C(C(O)=O)C(=O)C3=CC(F)=C2N1CCN(C)CC1 |
|---|
| InChI Identifier | InChI=1S/C18H20FN3O4/c1-10-9-26-17-14-11(16(23)12(18(24)25)8-22(10)14)7-13(19)15(17)21-5-3-20(2)4-6-21/h7-8,10H,3-6,9H2,1-2H3,(H,24,25)/t10-/m0/s1 |
|---|
| InChI Key | InChIKey=GSDSWSVVBLHKDQ-JTQLQIEISA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as quinoline carboxylic acids. These are quinolines in which the quinoline ring system is substituted by a carboxyl group at one or more positions. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Quinolines and derivatives |
|---|
| Sub Class | Quinoline carboxylic acids |
|---|
| Direct Parent | Quinoline carboxylic acids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Quinoline-3-carboxylic acid
- Fluoroquinolone
- N-arylpiperazine
- Aminoquinoline
- Haloquinoline
- Dihydroquinolone
- Benzoxazine
- Dihydroquinoline
- Pyridine carboxylic acid
- Pyridine carboxylic acid or derivatives
- Tertiary aliphatic/aromatic amine
- Dialkylarylamine
- N-alkylpiperazine
- N-methylpiperazine
- Alkyl aryl ether
- Benzenoid
- Pyridine
- Aryl fluoride
- Piperazine
- Aryl halide
- 1,4-diazinane
- Vinylogous amide
- Heteroaromatic compound
- Tertiary aliphatic amine
- Tertiary amine
- Amino acid or derivatives
- Amino acid
- Azacycle
- Oxacycle
- Carboxylic acid derivative
- Carboxylic acid
- Ether
- Monocarboxylic acid or derivatives
- Organonitrogen compound
- Organic nitrogen compound
- Amine
- Organic oxygen compound
- Organooxygen compound
- Organopnictogen compound
- Organic oxide
- Organofluoride
- Hydrocarbon derivative
- Organohalogen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Exogenous |
|---|
| Cellular Locations | |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | |
|---|
| Pathways | Not Available |
|---|
| Applications | |
|---|
| Biological Roles | |
|---|
| Chemical Roles | |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Appearance | Clear yellow to clear greenish-yellow liquid (7). |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Insoluble | | LogP | 2.1 |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | Deposition Date | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0006-2019000000-30d77332fe9feb70a82d | 2017-09-01 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-01dl-7009600000-d322709717558bd09e25 | 2017-10-06 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, N/A (Annotated) | splash10-00gm-1315975420-b0c4e6cdef799fda69b4 | 2012-07-25 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, N/A (Annotated) | splash10-0abc-3459425420-e8a4ecd2f8aea93fa660 | 2012-07-25 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, N/A (Annotated) | splash10-0h9p-3513893500-1d5c8069419fa2c37ebb | 2012-07-25 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-03di-0009000000-63138044688fa3ab0792 | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-02t9-0019000000-265eec70cbb77077fbfa | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-03di-1290000000-3ff2c315dfac97ded8c5 | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-0axr-1980000000-e31472e2487627c51cea | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-0adl-1930000000-3cd445f16118ecf3f610 | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - , positive | splash10-03di-0049000000-3cbab894a4b0d1165e7d | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - , positive | splash10-03di-2492000000-62b30ac9148b06791816 | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 75V, Positive | splash10-0axr-1980000000-aa52ed55d102e4d6142f | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 60V, Positive | splash10-03di-1190000000-77760aedde03dab8af8c | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 30V, Positive | splash10-02t9-0019000000-21613398a8dff5fc5e57 | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 15V, Positive | splash10-03di-0009000000-47e7a80ef7c8861677f6 | 2021-09-20 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-0009000000-6e3992e2c7f71e2e2d60 | 2017-07-25 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-02tc-0009000000-8e0f1b0fc3b80969c0dd | 2017-07-25 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0pi3-7097000000-363d82fc0e563a2423b0 | 2017-07-25 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-02t9-0009000000-bf579599f29511ea990c | 2017-07-26 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0670-0096000000-9d4af124e757cd0455bb | 2017-07-26 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-014r-0091000000-c5e349004a62406fb095 | 2017-07-26 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03dl-0009000000-c49f6f12144cd4de39cb | 2021-09-23 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0006-0009000000-7979df79be5019e8bd92 | 2021-09-23 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0udl-1079000000-68ac9a2c90e1aa665339 | 2021-09-23 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0019000000-ae9c58f7e3ad98591163 | 2021-09-24 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0002-0095000000-baf3b8295498cac74a7b | 2021-09-24 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, CD3OD, experimental) | Not Available | 2012-12-05 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, CD3OD, experimental) | Not Available | 2012-12-05 | View Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Ingestion; injection; skin contact (7).
Absorption of ofloxacin after single or multiple doses of 200 to 400 mg is predictable, and the amount of drug absorbed increases proportionately with the dose. |
|---|
| Mechanism of Toxicity | Levofloxacin inhibits bacterial type II topoisomerases, topoisomerase IV and DNA gyrase. Levofloxacin, like other fluoroquinolones, inhibits the A subunits of DNA gyrase, two subunits encoded by the gyrA gene. This results in strand breakage on a bacterial chromosome, supercoiling, and resealing; DNA replication and transcription is inhibited. |
|---|
| Metabolism | Mainly excreted as unchanged drug (87%); undergoes limited metabolism in humans. Levofloxacin is rapidly and essentially completely absorbed after oral administration. It is distributed into body tissues, especially in the skin and lung tissues. Levofloxacin is stereochemically stable in plasma and urine and does not invert metabolically to its enantiomer, D-ofloxacin. Levofloxacin undergoes limited metabolism in humans and is primarily excreted as unchanged drug in the urine. Following oral administration, approximately 87% of an administered dose was recovered as unchanged drug in urine within 48 hours, whereas less than 4% of the dose was recovered in feces in 72 hours. Less than 5% of an administered dose was recovered in the urine as the desmethyl and N-oxide metabolites, the only metabolites identified in humans. These metabolites have little relevant pharmacological activity. Levofloxacin is excreted largely as unchanged drug in the urine (6).
Route of Elimination: Mainly excreted as unchanged drug in the urine.
Half Life: 6-8 hours |
|---|
| Toxicity Values | LD50: 640 mg/kg (Oral, Rat) |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | No indication of carcinogenicity to humans (not listed by IARC). |
|---|
| Uses/Sources | For the treatment of bacterial conjunctivitis caused by susceptible strains of the following organisms: Corynebacterium species, Staphylococus aureus, Staphylococcus epidermidis, Streptococcus pneumoniae, Streptococcus (Groups C/F/G), Viridans group streptococci, Acinetobacter lwoffii, Haemophilus influenzae, Serratia marcescens. Levofloxacin is a synthetic chemotherapeutic agent used to treat severe and life threatening bacterial infection (6). |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Irreversible peripheral neuropathy, central nervous system toxicity, cardiovascular toxicity, tendon / articular toxicity, and hepatic toxicity. Toxic epidermal necrolysis, coagulation abnormalities and pancytopenia are other possible adverse effect of levofloxacin. Administration of commercial levofloxin (Levaquin), increased risk of tendinitis and tendon rupture in all ages. This adverse reaction most frequently involves the Achilles tendon (6, 7). |
|---|
| Symptoms | Side effects include disorientation, dizziness, drowsiness, hot and cold flashes, nausea, slurring of speech, swelling and numbness in the face. |
|---|
| Treatment | In the event of an acute overdosage, the stomach should be emptied. The patient should be observed and appropriate hydration maintained. Levofloxacin is not efficiently removed by hemodialysis or peritoneal dialysis. (6) |
|---|
| Normal Concentrations |
|---|
| Not Available |
|---|
| Abnormal Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB01137 |
|---|
| HMDB ID | HMDB01929 |
|---|
| PubChem Compound ID | 149096 |
|---|
| ChEMBL ID | CHEMBL33 |
|---|
| ChemSpider ID | 131410 |
|---|
| KEGG ID | C07660 |
|---|
| UniProt ID | Not Available |
|---|
| OMIM ID | |
|---|
| ChEBI ID | 63598 |
|---|
| BioCyc ID | Not Available |
|---|
| CTD ID | Not Available |
|---|
| Stitch ID | Levofloxacin |
|---|
| PDB ID | LFX |
|---|
| ACToR ID | Not Available |
|---|
| Wikipedia Link | Levofloxacin |
|---|
| References |
|---|
| Synthesis Reference | Valerie Niddam-Hildesheim, “Preparation of levofloxacin and forms thereof.” U.S. Patent US20030130507, issued July 10, 2003. |
|---|
| MSDS | Link |
|---|
| General References | - Wishart DS, Knox C, Guo AC, Cheng D, Shrivastava S, Tzur D, Gautam B, Hassanali M: DrugBank: a knowledgebase for drugs, drug actions and drug targets. Nucleic Acids Res. 2008 Jan;36(Database issue):D901-6. Epub 2007 Nov 29. [18048412 ]
- Norrby SR: Levofloxacin. Expert Opin Pharmacother. 1999 Nov;1(1):109-19. [11249554 ]
- Chow AT, Chen A, Lattime H, Morgan N, Wong F, Fowler C, Williams RR: Penetration of levofloxacin into skin tissue after oral administration of multiple 750 mg once-daily doses. J Clin Pharm Ther. 2002 Apr;27(2):143-50. [11975700 ]
- Bielecka-Grzela S, Klimowicz A: Evaluation of ofloxacin penetration into the skin after a single oral dose assessed by cutaneous microdialysis. Pol J Pharmacol. 2003 Jul-Aug;55(4):613-8. [14581720 ]
- Sreekumar A, Poisson LM, Rajendiran TM, Khan AP, Cao Q, Yu J, Laxman B, Mehra R, Lonigro RJ, Li Y, Nyati MK, Ahsan A, Kalyana-Sundaram S, Han B, Cao X, Byun J, Omenn GS, Ghosh D, Pennathur S, Alexander DC, Berger A, Shuster JR, Wei JT, Varambally S, Beecher C, Chinnaiyan AM: Metabolomic profiles delineate potential role for sarcosine in prostate cancer progression. Nature. 2009 Feb 12;457(7231):910-4. doi: 10.1038/nature07762. [19212411 ]
- Wikipedia. Levofloxacin. Last Updated 7 August 2009. [Link]
- RxList: The Internet Drug Index (2009). [Link]
- Pharmevo (2009). Spectrix. [Link]
- Drugs.com [Link]
|
|---|
| Gene Regulation |
|---|
| Up-Regulated Genes | Not Available |
|---|
| Down-Regulated Genes | Not Available |
|---|