| Record Information |
|---|
| Version | 2.0 |
|---|
| Creation Date | 2009-07-21 20:26:32 UTC |
|---|
| Update Date | 2014-12-24 20:25:51 UTC |
|---|
| Accession Number | T3D2743 |
|---|
| Identification |
|---|
| Common Name | Mitomycin |
|---|
| Class | Small Molecule |
|---|
| Description | Mitomycin is only found in individuals that have used or taken this drug. It is an antineoplastic antibiotic produced by Streptomyces caespitosus. It is one of the bi- or tri-functional alkylating agents causing cross-linking of DNA and inhibition of DNA synthesis. Mitomycin is activated in vivo to a bifunctional and trifunctional alkylating agent. Binding to DNA leads to cross-linking and inhibition of DNA synthesis and function. Mitomycin is cell cycle phase-nonspecific. |
|---|
| Compound Type | - Alkylating Agent
- Amide
- Amine
- Antibiotic, Antineoplastic
- Cross-Linking Reagent
- Drug
- Ester
- Ether
- Metabolite
- Nucleic Acid Synthesis Inhibitor
- Organic Compound
- Synthetic Compound
|
|---|
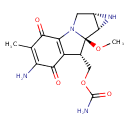
| Chemical Structure | |
|---|
| Synonyms | | Synonym | | 7-Amino-9alpha-methoxymitosane | | 7-Amino-9α-methoxymitosane | | Ametycine | | Mitamycin | | Mitocin-C | | Mitomycin C | | Mitosol | | Mitozytrex | | MMC | | Mutamycin |
|
|---|
| Chemical Formula | C15H18N4O5 |
|---|
| Average Molecular Mass | 334.327 g/mol |
|---|
| Monoisotopic Mass | 334.128 g/mol |
|---|
| CAS Registry Number | 50-07-7 |
|---|
| IUPAC Name | [(4S,6S,7R,8S)-11-amino-7-methoxy-12-methyl-10,13-dioxo-2,5-diazatetracyclo[7.4.0.0²,⁷.0⁴,⁶]trideca-1(9),11-dien-8-yl]methyl carbamate |
|---|
| Traditional Name | mitomycin c |
|---|
| SMILES | [H][C@]12CN3C4=C(C(=O)C(N)=C(C)C4=O)[C@@]([H])(COC(O)=N)[C@@]3(OC)[C@@]1([H])N2 |
|---|
| InChI Identifier | InChI=1S/C15H18N4O5/c1-5-9(16)12(21)8-6(4-24-14(17)22)15(23-2)13-7(18-13)3-19(15)10(8)11(5)20/h6-7,13,18H,3-4,16H2,1-2H3,(H2,17,22)/t6-,7+,13+,15-/m1/s1 |
|---|
| InChI Key | InChIKey=NWIBSHFKIJFRCO-WUDYKRTCSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as mitomycins. These are polycyclic compounds with a structure based on an aziridine ring linked to a 7-amino-6-methyl-cyclohexa[b]pyrrolizine-5,8-dione. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Indoles and derivatives |
|---|
| Sub Class | Indolequinones |
|---|
| Direct Parent | Mitomycins |
|---|
| Alternative Parents | |
|---|
| Substituents | - Mitomycin
- Indole
- Quinone
- Pyrrolizine
- 1,4-diazinane
- Piperazine
- Pyrrolidine
- Vinylogous amide
- Pyrroline
- Ketone
- Aziridine
- Carboximidic acid derivative
- Secondary aliphatic amine
- Enamine
- Azacycle
- Secondary amine
- Primary aliphatic amine
- Organic oxide
- Organopnictogen compound
- Organic oxygen compound
- Imine
- Carbonyl group
- Organic nitrogen compound
- Organonitrogen compound
- Organooxygen compound
- Hydrocarbon derivative
- Primary amine
- Amine
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Exogenous |
|---|
| Cellular Locations | |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Appearance | White powder. |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | >360°C | | Boiling Point | 534°C | | Solubility | Soluble (8430 mg/L) | | LogP | -0.4 |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | Deposition Date | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0k96-9032000000-addf881f05b59bd45e65 | 2017-09-01 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 40V, Positive | splash10-00l6-0950000000-a3b5f2c191b66d80a863 | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 20V, Positive | splash10-0006-0090000000-894659e6d592c7512847 | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0090000000-e936bf9eaf319819a3cd | 2021-09-20 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00kr-1039000000-d46d2d90cca8822c99bc | 2016-08-01 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0603-2094000000-94a0851dbf07fb7b2ede | 2016-08-01 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4l-9010000000-93ec12977feffeac422b | 2016-08-01 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-9012000000-22f59cef40bc61653f39 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0006-9000000000-0e9aba8bfa2e95134f7e | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9010000000-fdd39ddcbff28d118e30 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-0093000000-0f143a27a21adb246823 | 2021-09-22 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0036-3091000000-9147b389119a364d0a6a | 2021-09-22 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-4093000000-4df228a8d47a45ccd618 | 2021-09-22 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00dm-0090000000-378c12a5c718b619eafc | 2021-09-23 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0006-0092000000-1f40ea88019c1692b82c | 2021-09-23 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00kf-1095000000-4c654596a0b5503fe79d | 2021-09-23 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | Not Available | 2021-09-16 | View Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Intravesical, erratic, intravenous. |
|---|
| Mechanism of Toxicity | Mitomycin is activated in vivo to a bifunctional and trifunctional alkylating agent. Binding to DNA leads to cross-linking and inhibition of DNA synthesis and function. Mitomycin is cell cycle phase-nonspecific. |
|---|
| Metabolism | Primarily hepatic, some in various other tissues.
Route of Elimination: Approximately 10% of a dose of mitomycin is excreted unchanged in the urine.
Half Life: 8-48 min |
|---|
| Toxicity Values | LD50: 23 mg/kg (Oral, Mouse) (1)
LD50: 30 mg/kg (Oral, Rat) (1) |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | 2B, possibly carcinogenic to humans. (3) |
|---|
| Uses/Sources | For treatment of malignant neoplasm of lip, oral cavity, pharynx, digestive organs, peritoneum, female breast, and urinary bladder. Also used as an adjunct to ab externo glaucoma surgery. |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Mitomycin C causes delayed bone marrow toxicity and therefore it is usually administered at 6-weekly intervals. Prolonged use may result in permanent bone-marrow damage. It may also cause lung fibrosis and renal damage. |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Normal Concentrations |
|---|
| Not Available |
|---|
| Abnormal Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB00305 |
|---|
| HMDB ID | HMDB14450 |
|---|
| PubChem Compound ID | 5746 |
|---|
| ChEMBL ID | CHEMBL105 |
|---|
| ChemSpider ID | 5544 |
|---|
| KEGG ID | C06681 |
|---|
| UniProt ID | Not Available |
|---|
| OMIM ID | |
|---|
| ChEBI ID | 27504 |
|---|
| BioCyc ID | Not Available |
|---|
| CTD ID | Not Available |
|---|
| Stitch ID | Mitomycin |
|---|
| PDB ID | Not Available |
|---|
| ACToR ID | Not Available |
|---|
| Wikipedia Link | Mitomycin |
|---|
| References |
|---|
| Synthesis Reference | Leslie Jimenez, Zheng Wang, “Synthesis of mitomycin and its analogs.” U.S. Patent US5523411, issued June, 1972. |
|---|
| MSDS | Link |
|---|
| General References | - Wishart DS, Knox C, Guo AC, Cheng D, Shrivastava S, Tzur D, Gautam B, Hassanali M: DrugBank: a knowledgebase for drugs, drug actions and drug targets. Nucleic Acids Res. 2008 Jan;36(Database issue):D901-6. Epub 2007 Nov 29. [18048412 ]
- Drugs.com [Link]
- International Agency for Research on Cancer (2014). IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. [Link]
|
|---|
| Gene Regulation |
|---|
| Up-Regulated Genes | | Gene | Gene Symbol | Gene ID | Interaction | Chromosome | Details |
|---|
|
|---|
| Down-Regulated Genes | | Gene | Gene Symbol | Gene ID | Interaction | Chromosome | Details |
|---|
|
|---|