| Record Information |
|---|
| Version | 2.0 |
|---|
| Creation Date | 2009-07-21 20:28:25 UTC |
|---|
| Update Date | 2014-12-24 20:25:55 UTC |
|---|
| Accession Number | T3D2992 |
|---|
| Identification |
|---|
| Common Name | Econazole |
|---|
| Class | Small Molecule |
|---|
| Description | Econazole is only found in individuals that have used or taken this drug. It is a broad spectrum antimycotic with some action against Gram positive bacteria. It is used topically in dermatomycoses also orally and parenterally. [PubChem] Econazole interacts with 14-alpha demethylase, a cytochrome P-450 enzyme necessary to convert lanosterol to ergosterol. As ergosterol is an essential component of the fungal cell membrane, inhibition of its synthesis results in increased cellular permeability causing leakage of cellular contents. Econazole may also inhibit endogenous respiration, interact with membrane phospholipids, inhibit the transformation of yeasts to mycelial forms, inhibit purine uptake, and impair triglyceride and/or phospholipid biosynthesis. |
|---|
| Compound Type | - Amine
- Antifungal Agent
- Drug
- Ether
- Metabolite
- Organic Compound
- Organochloride
- Synthetic Compound
|
|---|
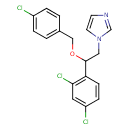
| Chemical Structure | |
|---|
| Synonyms | | Synonym | | (+-)-Econazole | | 1-(2,4-Dichloro-beta-((P-chlorobenzyl)oxy)phenethyl)imidazole | | Econazole Nitrate | | Ecostatin | | ECOZA | | Gyno-Pevaryl | | Ifenec | | Palavale | | Pevaryl | | Spectazole |
|
|---|
| Chemical Formula | C18H15Cl3N2O |
|---|
| Average Molecular Mass | 381.684 g/mol |
|---|
| Monoisotopic Mass | 380.025 g/mol |
|---|
| CAS Registry Number | 27220-47-9 |
|---|
| IUPAC Name | 1-{2-[(4-chlorophenyl)methoxy]-2-(2,4-dichlorophenyl)ethyl}-1H-imidazole |
|---|
| Traditional Name | econazole |
|---|
| SMILES | ClC1=CC=C(COC(CN2C=CN=C2)C2=C(Cl)C=C(Cl)C=C2)C=C1 |
|---|
| InChI Identifier | InChI=1/C18H15Cl3N2O/c19-14-3-1-13(2-4-14)11-24-18(10-23-8-7-22-12-23)16-6-5-15(20)9-17(16)21/h1-9,12,18H,10-11H2 |
|---|
| InChI Key | InChIKey=LEZWWPYKPKIXLL-UHFFFAOYNA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as benzylethers. These are aromatic ethers with the general formula ROCR' (R = alkyl, aryl; R'=benzene). |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Benzene and substituted derivatives |
|---|
| Sub Class | Benzylethers |
|---|
| Direct Parent | Benzylethers |
|---|
| Alternative Parents | |
|---|
| Substituents | - Benzylether
- 1,3-dichlorobenzene
- Chlorobenzene
- Halobenzene
- Aryl chloride
- Aryl halide
- N-substituted imidazole
- Imidazole
- Azole
- Heteroaromatic compound
- Ether
- Dialkyl ether
- Organoheterocyclic compound
- Azacycle
- Organooxygen compound
- Organonitrogen compound
- Organochloride
- Organohalogen compound
- Hydrocarbon derivative
- Organopnictogen compound
- Organic oxygen compound
- Organic nitrogen compound
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Exogenous |
|---|
| Cellular Locations | |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | |
|---|
| Biological Roles | |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Appearance | White powder. |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | 162°C | | Boiling Point | Not Available | | Solubility | 1.48e-03 g/L | | LogP | 5.5 |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | Deposition Date | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-004i-9341000000-4ffb0473a31cea3f8c18 | 2017-09-01 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - , positive | splash10-001i-0209000000-dd9a7887032ba981630f | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 75V, Positive | splash10-004i-1900000000-28f4c0046949a5e27ab1 | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 60V, Positive | splash10-004i-0900000000-f00842704123db9b0465 | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 30V, Positive | splash10-004i-0903000000-6c5549ea79b88c593f2e | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 15V, Positive | splash10-001i-0009000000-0e20351a1183f50f888e | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 90V, Positive | splash10-004i-5900000000-bb76504dc81de5c3b579 | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 45V, Positive | splash10-004i-0900000000-a667527ad6d687e25400 | 2021-09-20 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-0019000000-e4e35751739b91041ba8 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00m0-5169000000-ef3cb4a26c4fda6ee25d | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-014i-9310000000-88884e5d802b100711cd | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-1009000000-cef44f57a37b6b00c969 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-014i-9001000000-7c677fea699efb1a72f3 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-014l-9310000000-1d15b5e04ae4eb7ad414 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-2039000000-17f63aee73e32a973719 | 2021-10-11 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-001i-9551000000-33222b3a25a12499d2cb | 2021-10-11 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00lr-9000000000-cd84e8c432915c9cc869 | 2021-10-11 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-0009000000-7996a613e7d9dea8ceba | 2021-10-11 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001i-1219000000-1878634d92d58865370e | 2021-10-11 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00or-6910000000-84c9b556f80d88764610 | 2021-10-11 | View Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | After topical application to the skin of normal subjects, systemic absorption of econazole nitrate is extremely low. Although most of the applied drug remains on the skin surface, drug concentrations were found in the stratum corneum which, by far, exceeded the minimum inhibitory concentration for dermatophytes. |
|---|
| Mechanism of Toxicity | Econazole interacts with 14-α demethylase, a cytochrome P-450 enzyme necessary to convert lanosterol to ergosterol. As ergosterol is an essential component of the fungal cell membrane, inhibition of its synthesis results in increased cellular permeability causing leakage of cellular contents. Econazole may also inhibit endogenous respiration, interact with membrane phospholipids, inhibit the transformation of yeasts to mycelial forms, inhibit purine uptake, and impair triglyceride and/or phospholipid biosynthesis. |
|---|
| Metabolism | Hepatic. |
|---|
| Toxicity Values | LD50: 462 mg/kg (Oral, Mouse) (1)
LD50: 462 mg/kg (Oral, Rat) (1)
LD50: 668 mg/kg (Oral, Guinea pig) (1)
LD50: >160 mg/kg (Oral, Dog) (1) |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | No indication of carcinogenicity to humans (not listed by IARC). |
|---|
| Uses/Sources | For topical application in the treatment of tinea pedis, tinea cruris, and tinea corporis caused by Trichophyton rubrum, Trichophyton mentagrophytes, Trichophyton tonsurans, Microsporum canis, Microsporum audouini, Microsporum gypseum, and Epidermophyton floccosum, in the treatment of cutaneous candidiasis, and in the treatment of tinea versicolor. |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Normal Concentrations |
|---|
| Not Available |
|---|
| Abnormal Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB01127 |
|---|
| HMDB ID | HMDB15259 |
|---|
| PubChem Compound ID | 3198 |
|---|
| ChEMBL ID | CHEMBL808 |
|---|
| ChemSpider ID | 3086 |
|---|
| KEGG ID | C08068 |
|---|
| UniProt ID | Not Available |
|---|
| OMIM ID | |
|---|
| ChEBI ID | 4754 |
|---|
| BioCyc ID | Not Available |
|---|
| CTD ID | Not Available |
|---|
| Stitch ID | Econazole |
|---|
| PDB ID | Not Available |
|---|
| ACToR ID | Not Available |
|---|
| Wikipedia Link | Econazole |
|---|
| References |
|---|
| Synthesis Reference | Godefroi, E.F. and Heeres, J.; U.S. Patent 3,717,655; February 20,1973; assigned to Jansen Pharmaceutica NV, Belgium. |
|---|
| MSDS | Link |
|---|
| General References | - Wishart DS, Knox C, Guo AC, Cheng D, Shrivastava S, Tzur D, Gautam B, Hassanali M: DrugBank: a knowledgebase for drugs, drug actions and drug targets. Nucleic Acids Res. 2008 Jan;36(Database issue):D901-6. Epub 2007 Nov 29. [18048412 ]
|
|---|
| Gene Regulation |
|---|
| Up-Regulated Genes | Not Available |
|---|
| Down-Regulated Genes | Not Available |
|---|