| Record Information |
|---|
| Version | 2.0 |
|---|
| Creation Date | 2009-07-21 20:28:44 UTC |
|---|
| Update Date | 2014-12-24 20:25:56 UTC |
|---|
| Accession Number | T3D3033 |
|---|
| Identification |
|---|
| Common Name | Isocarboxazid |
|---|
| Class | Small Molecule |
|---|
| Description | Isocarboxazid is only found in individuals that have used or taken this drug. It is an MAO inhibitor that is effective in the treatment of major depression, dysthymic disorder, and atypical depression. It also is useful in the treatment of panic disorder and the phobic disorders. (From AMA, Drug Evaluations Annual, 1994, p311). Isocarboxazid works by irreversibly blocking the action of a chemical substance known as monoamine oxidase (MAO) in the nervous system. MAO subtypes A and B are involved in the metabolism of serotonin and catecholamine neurotransmitters such as epinephrine, norepinephrine, and dopamine. Isocarboxazid, as a nonselective MAO inhibitor, binds irreversibly to monoamine oxidase–A (MAO-A) and monoamine oxidase–B (MAO-B). The reduced MAO activity results in an increased concentration of these neurotransmitters in storage sites throughout the central nervous system (CNS) and sympathetic nervous system. This increased availability of one or more monoamines is the basis for the antidepressant activity of MAO inhibitors. |
|---|
| Compound Type | - Amide
- Amine
- Antidepressant
- Antidepressive Agent
- Drug
- Ester
- Hydrazine
- Metabolite
- Monoamine Oxidase Inhibitor
- Organic Compound
- Synthetic Compound
|
|---|
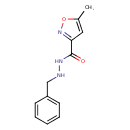
| Chemical Structure | |
|---|
| Synonyms | | Synonym | | Enerzer | | Isocarbonazid | | Isocarbossazide | | Isocarboxazida | | Isocarboxazide | | Isocarboxazidum | | Isocarboxyzid | | Marplan |
|
|---|
| Chemical Formula | C12H13N3O2 |
|---|
| Average Molecular Mass | 231.251 g/mol |
|---|
| Monoisotopic Mass | 231.101 g/mol |
|---|
| CAS Registry Number | 59-63-2 |
|---|
| IUPAC Name | N'-benzyl-5-methyl-1,2-oxazole-3-carbohydrazide |
|---|
| Traditional Name | isocarboxazid |
|---|
| SMILES | CC1=CC(=NO1)C(=O)NNCC1=CC=CC=C1 |
|---|
| InChI Identifier | InChI=1S/C12H13N3O2/c1-9-7-11(15-17-9)12(16)14-13-8-10-5-3-2-4-6-10/h2-7,13H,8H2,1H3,(H,14,16) |
|---|
| InChI Key | InChIKey=XKFPYPQQHFEXRZ-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as benzene and substituted derivatives. These are aromatic compounds containing one monocyclic ring system consisting of benzene. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Benzene and substituted derivatives |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Benzene and substituted derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Monocyclic benzene moiety
- Azole
- Isoxazole
- Heteroaromatic compound
- Carboxylic acid hydrazide
- Carboxylic acid derivative
- Organoheterocyclic compound
- Azacycle
- Oxacycle
- Organooxygen compound
- Organonitrogen compound
- Organic oxide
- Organopnictogen compound
- Organic nitrogen compound
- Organic oxygen compound
- Hydrocarbon derivative
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Exogenous |
|---|
| Cellular Locations | |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Appearance | White powder. |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | 105-106°C | | Boiling Point | Not Available | | Solubility | 1.6 mg/mL at 25°C | | LogP | 1.49 |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | Deposition Date | View |
|---|
| GC-MS | GC-MS Spectrum - EI-B (Non-derivatized) | splash10-0006-9600000000-952a7c1093fe532d1df3 | 2017-09-12 | View Spectrum | | GC-MS | GC-MS Spectrum - EI-B (Non-derivatized) | splash10-0006-9600000000-952a7c1093fe532d1df3 | 2018-05-18 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-052f-9510000000-c11f3aaa62df1be7ea3f | 2017-09-01 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-qTof , Positive | splash10-0006-9210100010-78b2f412502a142509e6 | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - , positive | splash10-0006-9210100010-78b2f412502a142509e6 | 2017-09-14 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-3290000000-9801781c24b97ecc0817 | 2016-08-01 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0006-9350000000-d8f9ca354f060f4330e2 | 2016-08-01 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0006-9100000000-6cedb1354fdeece131f6 | 2016-08-01 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-001i-1690000000-07a8545573baa300826f | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-001l-9640000000-077985063287a38f7095 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0kx0-9300000000-a6c0764f23b7adf2144e | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001l-6390000000-911dc29e6ed1ee470433 | 2021-10-11 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0006-9100000000-4896c74c701f3ec4b2fd | 2021-10-11 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0006-9200000000-cc7b99641fc7414fb559 | 2021-10-11 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-001i-0090000000-b203703a00eee615a207 | 2021-10-11 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-001l-9110000000-fc267c475e490b4f9f3c | 2021-10-11 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-000x-9100000000-b1b63bbca6e5cb19e8b8 | 2021-10-11 | View Spectrum | | MS | Mass Spectrum (Electron Ionization) | splash10-0006-9500000000-6ab30f52fc69889b3fa2 | 2014-09-20 | View Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Well absorbed from the gastrointestinal tract. |
|---|
| Mechanism of Toxicity | Isocarboxazid works by irreversibly blocking the action of a chemical substance known as monoamine oxidase (MAO) in the nervous system. MAO subtypes A and B are involved in the metabolism of serotonin and catecholamine neurotransmitters such as epinephrine, norepinephrine, and dopamine. Isocarboxazid, as a nonselective MAO inhibitor, binds irreversibly to monoamine oxidase–A (MAO-A) and monoamine oxidase–B (MAO-B). The reduced MAO activity results in an increased concentration of these neurotransmitters in storage sites throughout the central nervous system (CNS) and sympathetic nervous system. This increased availability of one or more monoamines is the basis for the antidepressant activity of MAO inhibitors. |
|---|
| Metabolism | Hepatic and rapid (by oxidation). |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | No indication of carcinogenicity to humans (not listed by IARC). |
|---|
| Uses/Sources | May be used to treat major depressive disorder. |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Signs of overdose include severe anxiety, confusion, convulsions, cool clammy skin, severe dizziness, severe drowsiness, fast and irregular pulse, fever, hallucinations, severe headache, high or low blood pressure, hyperactive reflexes, muscle stiffness, respiratory depression or failure, slowed reflexes, sweating, severe trouble in sleeping, and unusual irritability. |
|---|
| Treatment | General supportive measures should be used, along with immediate gastric lavage or emetics. If the latter are given, the danger of aspiration must be borne in mind. An adequate airway should be maintained, with supplemental oxygen if necessary. The mechanism by which amine-oxidase inhibitors produce hypotension is not fully understood, but there is evidence that these agents block the vascular bed response. Thus it is suggested that plasma may be of value in the management of this hypotension. Administration of pressor amines such as levarterenol bitartrate may be of limited value (note that their effects may be potentiated by Isocarboxazid). Continue treatment for several days until homeostasis is restored. Liver function studies are recommended during the 4 to 6 weeks after recovery, as well as the time of overdosage. |
|---|
| Normal Concentrations |
|---|
| Not Available |
|---|
| Abnormal Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB01247 |
|---|
| HMDB ID | HMDB15377 |
|---|
| PubChem Compound ID | 3759 |
|---|
| ChEMBL ID | CHEMBL1201168 |
|---|
| ChemSpider ID | 3628 |
|---|
| KEGG ID | Not Available |
|---|
| UniProt ID | Not Available |
|---|
| OMIM ID | |
|---|
| ChEBI ID | 775119 |
|---|
| BioCyc ID | Not Available |
|---|
| CTD ID | Not Available |
|---|
| Stitch ID | Isocarboxazid |
|---|
| PDB ID | Not Available |
|---|
| ACToR ID | Not Available |
|---|
| Wikipedia Link | Isocarboxazid |
|---|
| References |
|---|
| Synthesis Reference | U.S. Patent 2,908,688. |
|---|
| MSDS | T3D3033.pdf |
|---|
| General References | - Kennedy SH, Piran N, Warsh JJ, Prendergast P, Mainprize E, Whynot C, Garfinkel PE: A trial of isocarboxazid in the treatment of bulimia nervosa. J Clin Psychopharmacol. 1988 Dec;8(6):391-6. [3069879 ]
- Drugs.com [Link]
|
|---|
| Gene Regulation |
|---|
| Up-Regulated Genes | Not Available |
|---|
| Down-Regulated Genes | Not Available |
|---|