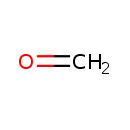| Record Information |
|---|
| Version | 2.0 |
|---|
| Creation Date | 2009-03-06 18:58:21 UTC |
|---|
| Update Date | 2014-12-24 20:21:24 UTC |
|---|
| Accession Number | T3D0244 |
|---|
| Identification |
|---|
| Common Name | Formaldehyde |
|---|
| Class | Small Molecule |
|---|
| Description | Formaldehyde is a highly reactive aldehyde gas formed by oxidation or incomplete combustion of hydrocarbons. In solution, it has a wide range of uses: in the manufacture of resins and textiles, as a disinfectant, and as a laboratory fixative or preservative. Formaldehyde solution (formalin) is considered a hazardous compound, and its vapor toxic. (From Reynolds, Martindale The Extra Pharmacopoeia, 30th ed, p717) -- Pubchem; The chemical compound formaldehyde (also known as methanal), is a gas with a pungent smell. It is the simplest aldehyde. Its chemical formula is H2CO. Formaldehyde was first synthesized by the Russian chemist Aleksandr Butlerov in 1859 but was conclusively identified by August Wilhelm van Hofmann in 1867. Although formaldehyde is a gas at room temperature, it is readily soluble in water, and it is most commonly sold as a 37% solution in water called by trade names such as formalin or formol. In water, formaldehyde polymerizes, and formalin actually contains very little formaldehyde in the form of H2CO monomer. Usually, these solutions contain a few percent methanol to limit the extent of polymerization. Formaldehyde exhibits most of the general chemical properties of the aldehydes, except that is generally more reactive than other aldehydes. Formaldehyde is a potent electrophile. It can participate in electrophilic aromatic substitution reactions with aromatic compounds and can undergo electrophilic addition reactions with alkenes. In the presence of basic catalysts, formaldehyde undergoes a Cannizaro reaction to produce formic acid and methanol. Because formaldehyde resins are used in many construction materials, including plywood, carpet, and spray-on insulating foams, and because these resins slowly give off formaldehyde over time, formaldehyde is one of the more common indoor air pollutants. At concentrations above 0.1 mg/kg in air, inhaled formaldehyde can irritate the eyes and mucous membranes, resulting in watery eyes, headache, a burning sensation in the throat, and difficulty breathing. -- Wikipedia. |
|---|
| Compound Type | - Aldehyde
- Cigarette Toxin
- Cosmetic Toxin
- Disinfectant
- Fixative
- Food Toxin
- Household Toxin
- Industrial Precursor/Intermediate
- Industrial/Workplace Toxin
- Lachrymator
- Metabolite
- Natural Compound
- Organic Compound
- Pollutant
|
|---|
| Chemical Structure | |
|---|
| Synonyms | | Synonym | | Fannoform | | Formalin | | Formalina | | Formaline | | Formalith | | Formic aldehyde | | Formol | | Methaldehyde | | Methanal | | Methyl aldehyde | | Methylene glycol | | Methylene oxide | | Oxomethane | | Oxomethylene | | Oxymethylene | | Paraform | | Paraformaldehyde |
|
|---|
| Chemical Formula | CH2O |
|---|
| Average Molecular Mass | 30.026 g/mol |
|---|
| Monoisotopic Mass | 30.011 g/mol |
|---|
| CAS Registry Number | 50-00-0 |
|---|
| IUPAC Name | formaldehyde |
|---|
| Traditional Name | formaldehyde |
|---|
| SMILES | C=O |
|---|
| InChI Identifier | InChI=1S/CH2O/c1-2/h1H2 |
|---|
| InChI Key | InChIKey=WSFSSNUMVMOOMR-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as carbonyl compounds. These are organic compounds containing a carbonyl group, with the general structure RC(=O)R', where R=organyl, R'=H, N, O, organyl group or halide group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Carbonyl compounds |
|---|
| Direct Parent | Carbonyl compounds |
|---|
| Alternative Parents | |
|---|
| Substituents | - Organic oxide
- Hydrocarbon derivative
- Carbonyl group
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Exogenous |
|---|
| Cellular Locations | - Cytoplasm
- Extracellular
- Lysosome
- Mitochondria
- Peroxisome
|
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | - Adipose Tissue
- Adrenal Cortex
- Adrenal Gland
- Adrenal Medulla
- Bladder
- Bone Marrow
- Brain
- Fibroblasts
- Gonads
- Intestine
- Kidney
- Liver
- Lung
- Muscle
- Myelin
- Nerve Cells
- Neuron
- Pancreas
- Placenta
- Platelet
- Prostate
- Skeletal Muscle
- Skin
|
|---|
| Pathways | |
|---|
| Applications | |
|---|
| Biological Roles | |
|---|
| Chemical Roles | |
|---|
| Physical Properties |
|---|
| State | Liquid |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | -92°C | | Boiling Point | Not Available | | Solubility | 400 mg/mL at 20°C [PICKRELL,JA et al. (1983)] | | LogP | 0.35 |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | Deposition Date | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-001i-9000000000-2863df3346e6e0796248 | 2016-09-22 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-9000000000-793a330f9e6c7661e86d | 2015-04-24 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001i-9000000000-793a330f9e6c7661e86d | 2015-04-24 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-001i-9000000000-793a330f9e6c7661e86d | 2015-04-24 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-9000000000-5ea4ad4bdb0565b737bb | 2015-04-25 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-004i-9000000000-5ea4ad4bdb0565b737bb | 2015-04-25 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-004i-9000000000-5ea4ad4bdb0565b737bb | 2015-04-25 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-9000000000-1534590d4d03d9b463f5 | 2021-09-25 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-004i-9000000000-1534590d4d03d9b463f5 | 2021-09-25 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-004i-9000000000-1534590d4d03d9b463f5 | 2021-09-25 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-9000000000-eaca5f454fe5238ba078 | 2021-09-25 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001i-9000000000-eaca5f454fe5238ba078 | 2021-09-25 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-001i-9000000000-eaca5f454fe5238ba078 | 2021-09-25 | View Spectrum | | MS | Mass Spectrum (Electron Ionization) | splash10-004i-9000000000-58b4df664ee160d1f943 | 2014-09-20 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, TMS, experimental) | Not Available | 2014-09-20 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, D2O, experimental) | Not Available | 2016-10-22 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, D2O, experimental) | Not Available | 2016-10-22 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | Not Available | 2021-09-29 | View Spectrum | | 2D NMR | [1H, 1H]-TOCSY. Unexported temporarily by An Chi on Oct 15, 2021 until json or nmrML file is generated. 2D NMR Spectrum (experimental) | Not Available | 2012-12-04 | View Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Oral (26) ; inhalation (26) ; dermal (26) |
|---|
| Mechanism of Toxicity | It is likely that formaldehyde toxicity occurs when intracellular levels saturate formaldehyde dehydrogenase activity, allowing the unmetabolized intact molecule to exert its effects. Formaldehyde is known to form cross links between protein and DNA and undergo metabolic incorporation into macromolecules (DNA, RNA, and proteins). (26) |
|---|
| Metabolism | Formaldehyde may be absorbed following inhalation, oral, or dermal exposure. It is an essential metabolic intermediate in all cells and is produced during the normal metabolism of serine, glycine, methionine, and choline and also by the demethylation of N-, S-, and O-methyl compounds. Exogenous formaldehyde is metabolized to formate by the enzyme formaldehyde dehydrogenase at the initial site of contact. After oxidation of formaldehyde to formate, the carbon atom is further oxidized to carbon dioxide or incorporated into purines, thymidine, and amino acids via tetrahydrofolatedependent one-carbon biosynthetic pathways. Formaldehyde is not stored in the body and is excreted in the urine (primarily as formic acid), incorporated into other cellular molecules, or exhaled as carbon dioxide. (26) |
|---|
| Toxicity Values | LD50: 300 mg/kg (Subcutaneous, Mouse) (22)
LD50: 42 mg/kg (Oral, Mouse) (22)
LD50: 87 mg/kg (Intravenous, Rat) (22)
LD50: 16 mg/kg (Intraperitoneal, Mouse) (23)
LC50: 0.414 mg/L over 4 hours (Inhalation, Mouse) (1) |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | 1, carcinogenic to humans. (25) |
|---|
| Uses/Sources | Formaldehyde is used in the production of fertilizer, paper, plywood, and urea-formaldehyde resins. It is also also used as a preservative in some foods and in many products used around the house, such as antiseptics, medicines, and cosmetics. (26) |
|---|
| Minimum Risk Level | Acute Inhalation: 0.04 ppm (24)
Intermediate Inhalation: 0.03 ppm (24)
Chronic Inhalation: 0.008 ppm (24)
Intermediate Oral: 0.3 mg/kg/day (24)
Chronic Oral: 0.2 mg/kg/day (24) |
|---|
| Health Effects | Drinking large amounts of formaldehyde can cause severe pain, vomiting, coma, and possible death. Formaldehyde is also a known human carcinogen. (26) |
|---|
| Symptoms | Low levels of formaldehyde can cause irritation of the eyes, nose, throat, and skin. (26) |
|---|
| Treatment | Not Available |
|---|
| Normal Concentrations |
|---|
| Not Available |
|---|
| Abnormal Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB03843 |
|---|
| HMDB ID | HMDB01426 |
|---|
| PubChem Compound ID | 712 |
|---|
| ChEMBL ID | CHEMBL1255 |
|---|
| ChemSpider ID | 692 |
|---|
| KEGG ID | C00067 |
|---|
| UniProt ID | Not Available |
|---|
| OMIM ID | |
|---|
| ChEBI ID | 16842 |
|---|
| BioCyc ID | FORMALDEHYDE |
|---|
| CTD ID | D005557 |
|---|
| Stitch ID | Formaldehyde |
|---|
| PDB ID | FOR |
|---|
| ACToR ID | 647 |
|---|
| Wikipedia Link | Formaldehyde |
|---|
| References |
|---|
| Synthesis Reference | Walter Hasselman, Jr., “Process for the preparation of phenol-urea-formaldehyde condensation products.” U.S. Patent US4345061, issued August, 1926. |
|---|
| MSDS | Link |
|---|
| General References | - Seifert J, Pewnim T: Alteration of mice L-tryptophan metabolism by the organophosphorous acid triester diazinon. Biochem Pharmacol. 1992 Dec 1;44(11):2243-50. [1282004 ]
- Lu CY, Chou AK, Wu CL, Yang CH, Chen JT, Wu PC, Lin SH, Muhammad R, Yang LC: Gene-gun particle with pro-opiomelanocortin cDNA produces analgesia against formalin-induced pain in rats. Gene Ther. 2002 Aug;9(15):1008-14. [12101431 ]
- Yang G, Ayala G, De Marzo A, Tian W, Frolov A, Wheeler TM, Thompson TC, Harper JW: Elevated Skp2 protein expression in human prostate cancer: association with loss of the cyclin-dependent kinase inhibitor p27 and PTEN and with reduced recurrence-free survival. Clin Cancer Res. 2002 Nov;8(11):3419-26. [12429629 ]
- Castrillon DH, Sun D, Weremowicz S, Fisher RA, Crum CP, Genest DR: Discrimination of complete hydatidiform mole from its mimics by immunohistochemistry of the paternally imprinted gene product p57KIP2. Am J Surg Pathol. 2001 Oct;25(10):1225-30. [11688455 ]
- Marinkovic S, Gibo H, Zelic O, Nikodijevic I: The neurovascular relationships and the blood supply of the trochlear nerve: surgical anatomy of its cisternal segment. Neurosurgery. 1996 Jan;38(1):161-9. [8747965 ]
- Pagani S, Mirtella D, Mencarelli R, Rodriguez D, Cingolani M: Postmortem distribution of sildenafil in histological material. J Anal Toxicol. 2005 May-Jun;29(4):254-7. [15975255 ]
- Heck Hd, Casanova M: The implausibility of leukemia induction by formaldehyde: a critical review of the biological evidence on distant-site toxicity. Regul Toxicol Pharmacol. 2004 Oct;40(2):92-106. [15450713 ]
- Shinkai M, Shinkai T, Puri P, Stringer MD: Elevated expression of IL2 is associated with increased infiltration of CD8+ T cells in biliary atresia. J Pediatr Surg. 2006 Feb;41(2):300-5. [16481239 ]
- Panasiuk A, Prokopowicz D, Dzieciol J: p53 protein expression in chronic hepatitis C; effect of interferon alpha 2b therapy. Hepatogastroenterology. 2005 Jul-Aug;52(64):1176-9. [16001656 ]
- van Dijk WR, van Haperen MJ, Stefanko SZ, van der Kamp AW: Monoclonal antibody selectively reactive with myelin sheaths of the peripheral nervous system in paraffin-embedded material. Acta Neuropathol. 1986;71(3-4):311-5. [2432751 ]
- Shakleya DM, Kraner JC, Kaplan JA, Gannett PM, Callery PS: Identification of N,N-dimethylamphetamine formed by methylation of methamphetamine in formalin-fixed liver tissue by multistage mass spectrometry. Forensic Sci Int. 2006 Mar 10;157(2-3):87-92. [15893897 ]
- Burke AP, Mont E, Kolodgie F, Virmani R: Thrombotic thrombocytopenic purpura causing rapid unexpected death: value of CD61 immunohistochemical staining in diagnosis. Cardiovasc Pathol. 2005 May-Jun;14(3):150-5. [15914300 ]
- Visee S, Soltner C, Rialland X, Machet MC, Loussouarn D, Milinkevitch S, Pasco-Papon A, Mercier P, Rousselet MC: Supratentorial primitive neuroectodermal tumours of the brain: multidirectional differentiation does not influence prognosis. A clinicopathological report of 18 patients. Histopathology. 2005 Apr;46(4):403-12. [15810952 ]
- Sriram S, Ljunggren-Rose A, Yao SY, Whetsell WO Jr: Detection of chlamydial bodies and antigens in the central nervous system of patients with multiple sclerosis. J Infect Dis. 2005 Oct 1;192(7):1219-28. Epub 2005 Sep 2. [16136465 ]
- Wilfredo Canchis P, Gonzalez SA, Isabel Fiel M, Chiriboga L, Yee H, Edlin BR, Jacobson IM, Talal AH: Hepatocyte proliferation in chronic hepatitis C: correlation with degree of liver disease and serum alpha-fetoprotein. Liver Int. 2004 Jun;24(3):198-203. [15189269 ]
- Zeldin DC, Foley J, Boyle JE, Moomaw CR, Tomer KB, Parker C, Steenbergen C, Wu S: Predominant expression of an arachidonate epoxygenase in islets of Langerhans cells in human and rat pancreas. Endocrinology. 1997 Mar;138(3):1338-46. [9048644 ]
- Moore GR, Leung E, MacKay AL, Vavasour IM, Whittall KP, Cover KS, Li DK, Hashimoto SA, Oger J, Sprinkle TJ, Paty DW: A pathology-MRI study of the short-T2 component in formalin-fixed multiple sclerosis brain. Neurology. 2000 Nov 28;55(10):1506-10. [11094105 ]
- Koscielny J, Aslan T, Meyer O, Kiesewetter H, Jung F, Mrowietz C, Latza R: Use of the platelet reactivity index by Grotemeyer, platelet function analyzer, and retention test Homburg to monitor therapy with antiplatelet drugs. Semin Thromb Hemost. 2005;31(4):464-9. [16149025 ]
- Nader N, Raverot G, Emptoz-Bonneton A, Dechaud H, Bonnay M, Baudin E, Pugeat M: Mitotane has an estrogenic effect on sex hormone-binding globulin and corticosteroid-binding globulin in humans. J Clin Endocrinol Metab. 2006 Jun;91(6):2165-70. Epub 2006 Mar 21. [16551731 ]
- North WG, Memoli VA, Keegan BP: Immunohistochemical detection of NRSA on small cell lung cancer with a monoclonal antibody (MAG-1) that recognizes the carboxyl terminus of provasopressin. Appl Immunohistochem Mol Morphol. 2005 Dec;13(4):363-6. [16280667 ]
- Katsetos CD, Jami MM, Krishna L, Jackson R, Patchefsky AS, Cooper HS: Novel immunohistochemical localization of 28,000 molecular-weight (Mr) calcium binding protein (calbindin-D28k) in enterochromaffin cells of the human appendix and neuroendocrine tumors (carcinoids and small-cell carcinomas) of the midgut and foregut. Arch Pathol Lab Med. 1994 Jun;118(6):633-9. [8204010 ]
- Lewis RJ Sr. (ed) (2004). Sax's Dangerous Properties of Industrial Materials. 11th Edition. Hoboken, NJ: Wiley-Interscience, Wiley & Sons, Inc.
- Bingham, E, Cohrssen, B, and Powell, CH (2001). Patty's Toxicology Volumes 1-9. 5th ed. New York, N.Y: John Wiley & Sons.
- ATSDR - Agency for Toxic Substances and Disease Registry (2001). Minimal Risk Levels (MRLs) for Hazardous Substances. U.S. Public Health Service in collaboration with U.S. Environmental Protection Agency (EPA). [Link]
- International Agency for Research on Cancer (2014). IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. [Link]
- ATSDR - Agency for Toxic Substances and Disease Registry (1999). Toxicological profile for formaldehyde. U.S. Public Health Service in collaboration with U.S. Environmental Protection Agency (EPA). [Link]
|
|---|
| Gene Regulation |
|---|
| Up-Regulated Genes | | Gene | Gene Symbol | Gene ID | Interaction | Chromosome | Details |
|---|
|
|---|
| Down-Regulated Genes | | Gene | Gene Symbol | Gene ID | Interaction | Chromosome | Details |
|---|
|
|---|