| Record Information |
|---|
| Version | 2.0 |
|---|
| Creation Date | 2009-03-06 18:58:10 UTC |
|---|
| Update Date | 2014-12-24 20:21:13 UTC |
|---|
| Accession Number | T3D0145 |
|---|
| Identification |
|---|
| Common Name | Parathion |
|---|
| Class | Small Molecule |
|---|
| Description | Parathion is a highly toxic cholinesterase inhibitor that is used as an acaricide and as an insecticide, generally applied by spraying, most often to cotton, rice, and fruit trees. Also called parathion-ethyl or diethyl parathion, parathion is an organophosphate compound. As parathion is highly toxic to non-target organisms, its use is banned or restricted in many areas. (17) |
|---|
| Compound Type | - Aromatic Hydrocarbon
- Ester
- Food Toxin
- Metabolite
- Organic Compound
- Organophosphate
- Pesticide
- Pollutant
- Synthetic Compound
|
|---|
| Chemical Structure | |
|---|
| Synonyms | | Synonym | | Alkron | | Alleron | | Aphamite | | Aqua 9-Parathion | | Aralo | | Bladan | | Bladen | | Corothion | | Corthione | | Danthion | | Deoxynucleoside 5'-triphosphate | | Diethyl 4-nitrophenyl phosphorothionate | | Diethyl O-p-nitrophenyl phosphorothioate | | Diethyl O-p-nitrophenyl phosphorothioic acid | | Diethyl p-nitrophenyl thionophosphate | | Diethyl p-nitrophenyl thiophosphate | | Diethyl para-nitrophenol thiophosphate | | Diethyl-p-nitrophenyl monothiophosphate | | Diethylparathion | | Drexel parathion 8E | | Durathion | | Ecatox | | Ethlon | | Ethyl parathion | | Ethyl parathion (O,O-diethyl-O-p-nitrophenylthiophosphate) | | Etilon | | Foliclal | | Folidol | | Folidol e e 605 | | Folidol e605 | | Folidol oil | | Fosfermo | | Fosferno | | Fosferno 50 | | Fosfex | | Fosfive | | Fosova | | Fostern | | Fostox | | Gearphos | | Genithion | | Kolphos | | Kypthion | | Lethalaire g-54 | | Lirothion | | Murfos | | Niran | | Nitrostygmine | | Niuif-100 | | Nourithion | | O,O-Diethyl O-p-nitrophenyl phosphorothioate | | O,O-Diethyl O-p-nitrophenyl phosphorothioic acid | | O,O-Diethyl-O-(p-nitrophenyl)thionophosphate | | Oleofos 20 | | Oleoparaphene | | Oleoparathion | | OMS 19 | | Orthophos | | p-Nitrophenol O-ester with O,O-diethylphosphorothioate | | p-Nitrophenol O-ester with O,O-diethylphosphorothioic acid | | PAC | | Pacol | | Panthion | | Paradust | | Paramar | | Paramar 50 | | Paraphos | | Parathene | | Parathion-E | | Parawet | | Parthion | | Penncap e | | Pestox plus | | Pethion | | Phoskil | | Phosphemol | | Phosphenol | | Phosphorothioate | | Phosphorothioic acid | | Phosphorothioic acid O,O-diethyl-O-(4-nitrophenyl) ester | | Phosphostigmine | | Rhodiasol | | Rhodiatox | | Rhodiatrox | | Selephos | | Soprathion | | Stabilized ethyl parathion | | Stathion | | Strathion | | Sulfos | | Super rodiatox | | T-47 | | Thiomex | | Thionspray No.84 | | Thiophos 3422 | | Tiofos | | Vapophos | | Viran | | Vitrex |
|
|---|
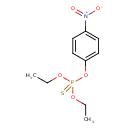
| Chemical Formula | C10H14NO5PS |
|---|
| Average Molecular Mass | 291.261 g/mol |
|---|
| Monoisotopic Mass | 291.033 g/mol |
|---|
| CAS Registry Number | 56-38-2 |
|---|
| IUPAC Name | O,O-diethyl O-4-nitrophenyl phosphorothioate |
|---|
| Traditional Name | bladen |
|---|
| SMILES | CCOP(=S)(OCC)OC1=CC=C(C=C1)N(=O)=O |
|---|
| InChI Identifier | InChI=1S/C10H14NO5PS/c1-3-14-17(18,15-4-2)16-10-7-5-9(6-8-10)11(12)13/h5-8H,3-4H2,1-2H3 |
|---|
| InChI Key | InChIKey=LCCNCVORNKJIRZ-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as phenyl thiophosphates. These are organothiophosphorus compounds that contain a thiophosphoric acid O-esterified with a phenyl group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Organic thiophosphoric acids and derivatives |
|---|
| Sub Class | Thiophosphoric acid esters |
|---|
| Direct Parent | Phenyl thiophosphates |
|---|
| Alternative Parents | |
|---|
| Substituents | - Phenyl thiophosphate
- Nitrobenzene
- Phenoxy compound
- Nitroaromatic compound
- Thiophosphate triester
- Monocyclic benzene moiety
- Benzenoid
- C-nitro compound
- Organic nitro compound
- Organic oxoazanium
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Allyl-type 1,3-dipolar organic compound
- Organic nitrogen compound
- Hydrocarbon derivative
- Organooxygen compound
- Organonitrogen compound
- Organic oxide
- Organopnictogen compound
- Organic oxygen compound
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Exogenous |
|---|
| Cellular Locations | |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | |
|---|
| Biological Roles | |
|---|
| Chemical Roles | |
|---|
| Physical Properties |
|---|
| State | Liquid |
|---|
| Appearance | White crystals. |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | 6.1°C | | Boiling Point | Not Available | | Solubility | 0.011 mg/mL | | LogP | 3.83 |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | Deposition Date | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-03fr-1390000000-256208006a2b1561db1b | 2017-09-01 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-01p9-0090000000-fe8742077ddcdaa84280 | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-000i-0190000000-af44a385ea22b6326819 | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-000l-1940000000-218556da13b43e16e2bf | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-01vo-2910000000-4a40f241595d8b5628d0 | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-01vo-4900000000-c58ec980fa6b4210133c | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-01ox-6900000000-80ce5f0f59ebc4a62681 | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 90V, Positive | splash10-01ox-6900000000-0ae225ae5aa0b11161e3 | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 15V, Positive | splash10-01p9-0090000000-699baba652581ceae2be | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 45V, Positive | splash10-000l-1940000000-15d927af1afee2c09028 | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 30V, Positive | splash10-000i-0190000000-13506519f0e449b52678 | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 60V, Positive | splash10-01vo-2910000000-e81debcb63112f1a8187 | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 75V, Positive | splash10-01vo-4900000000-697208a779d2fefe32fd | 2021-09-20 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-1090000000-e7daa1435a3c8202b8dc | 2016-08-01 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00lr-1090000000-0ee220c0c9ec67d726bb | 2016-08-01 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-004j-9720000000-be163305078ac89a7681 | 2016-08-01 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-0390000000-3cdf27289e65564f2028 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0006-0490000000-9b32c86de4c27c33c405 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-000f-1940000000-6f5772e73d0e7958d6cb | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0090000000-9585d70a69dd82c677f0 | 2021-09-23 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01pc-0090000000-7f509a774ae9d3a5ac84 | 2021-09-23 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0udi-9340000000-b44b4f76ff61f463d2db | 2021-09-23 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-0090000000-f2d3ba63a2de010595cf | 2021-09-24 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-0290000000-b84c6ef99ca616492499 | 2021-09-24 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-006t-7910000000-fc58d956a883b5d8b5ca | 2021-09-24 | View Spectrum | | MS | Mass Spectrum (Electron Ionization) | splash10-0a74-8940000000-4bf55c77de23f747eb80 | 2014-09-20 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 600 MHz, CDCl3, experimental) | Not Available | 2012-12-04 | View Spectrum | | 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, CDCl3, experimental) | Not Available | 2012-12-05 | View Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Oral(17) ; inhalation (17) ; dermal (17). |
|---|
| Mechanism of Toxicity | Parathion is a cholinesterase or acetylcholinesterase (AChE) inhibitor. A cholinesterase inhibitor (or 'anticholinesterase') suppresses the action of acetylcholinesterase. Because of its essential function, chemicals that interfere with the action of acetylcholinesterase are potent neurotoxins, causing excessive salivation and eye-watering in low doses, followed by muscle spasms and ultimately death. Nerve gases and many substances used in insecticides have been shown to act by binding a serine in the active site of acetylcholine esterase, inhibiting the enzyme completely. Acetylcholine esterase breaks down the neurotransmitter acetylcholine, which is released at nerve and muscle junctions, in order to allow the muscle or organ to relax. The result of acetylcholine esterase inhibition is that acetylcholine builds up and continues to act so that any nerve impulses are continually transmitted and muscle contractions do not stop. Among the most common acetylcholinesterase inhibitors are phosphorus-based compounds, which are designed to bind to the active site of the enzyme. The structural requirements are a phosphorus atom bearing two lipophilic groups, a leaving group (such as a halide or thiocyanate), and a terminal oxygen. |
|---|
| Metabolism | Metabolism of organophosphates occurs principally by oxidation, by hydrolysis via esterases and by reaction with glutathione. Demethylation and glucuronidation may also occur. Oxidation of organophosphorus pesticides may result in moderately toxic products. In general, phosphorothioates are not directly toxic but require oxidative metabolism to the proximal toxin. The glutathione transferase reactions produce products that are, in most cases, of low toxicity. Paraoxonase (PON1) is a key enzyme in the metabolism of organophosphates. PON1 can inactivate some organophosphates through hydrolysis. PON1 hydrolyzes the active metabolites in several organophosphates insecticides as well as, nerve agents such as soman, sarin, and VX. The presence of PON1 polymorphisms causes there to be different enzyme levels and catalytic efficiency of this esterase, which in turn suggests that different individuals may be more susceptible to the toxic effect of organophosphate exposure. |
|---|
| Toxicity Values | LD50: 2 mg/kg (Oral, Rat) (13)
LD50: 6800 ug/kg (Dermal, Rat) (13)
LD05: 3600 ug/kg (Intraperitoneal, Rat) (13)
LD50: 6 mg/kg (Intramuscular, Rat) (13)
LC50: 84.0 mg (Inhalation, Rat) (4) |
|---|
| Lethal Dose | 7143 mg/kg for an adult human. (14) |
|---|
| Carcinogenicity (IARC Classification) | 3, not classifiable as to its carcinogenicity to humans. (16) |
|---|
| Uses/Sources | Parathion is an insecticide and acaricide that is generally applied by spraying, most often to cotton, rice, and fruit trees. (17) |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Acute exposure to cholinesterase inhibitors can cause a cholinergic crisis characterized by severe nausea/vomiting, salivation, sweating, bradycardia, hypotension, collapse, and convulsions. Increasing muscle weakness is a possibility and may result in death if respiratory muscles are involved. Accumulation of ACh at motor nerves causes overstimulation of nicotinic expression at the neuromuscular junction. When this occurs symptoms such as muscle weakness, fatigue, muscle cramps, fasciculation, and paralysis can be seen. When there is an accumulation of ACh at autonomic ganglia this causes overstimulation of nicotinic expression in the sympathetic system. Symptoms associated with this are hypertension, and hypoglycemia. Overstimulation of nicotinic acetylcholine receptors in the central nervous system, due to accumulation of ACh, results in anxiety, headache, convulsions, ataxia, depression of respiration and circulation, tremor, general weakness, and potentially coma. When there is expression of muscarinic overstimulation due to excess acetylcholine at muscarinic acetylcholine receptors symptoms of visual disturbances, tightness in chest, wheezing due to bronchoconstriction, increased bronchial secretions, increased salivation, lacrimation, sweating, peristalsis, and urination can occur. Certain reproductive effects in fertility, growth, and development for males and females have been linked specifically to organophosphate pesticide exposure. Most of the research on reproductive effects has been conducted on farmers working with pesticides and insecticdes in rural areas. In females menstrual cycle disturbances, longer pregnancies, spontaneous abortions, stillbirths, and some developmental effects in offspring have been linked to organophosphate pesticide exposure. Prenatal exposure has been linked to impaired fetal growth and development. Neurotoxic effects have also been linked to poisoning with OP pesticides causing four neurotoxic effects in humans: cholinergic syndrome, intermediate syndrome, organophosphate-induced delayed polyneuropathy (OPIDP), and chronic organophosphate-induced neuropsychiatric disorder (COPIND). These syndromes result after acute and chronic exposure to OP pesticides. |
|---|
| Symptoms | Parathion exposure can result in headaches, convulsions, poor vision, vomiting, abdominal pain, severe diarrhea, unconsciousness, tremor, dyspnea, and finally lung-edema as well as respiratory arrest. (17) |
|---|
| Treatment | If the compound has been ingested, rapid gastric lavage should be performed using 5% sodium bicarbonate. For skin contact, the skin should be washed with soap and water. If the compound has entered the eyes, they should be washed with large quantities of isotonic saline or water. In serious cases, atropine and/or pralidoxime should be administered. Anti-cholinergic drugs work to counteract the effects of excess acetylcholine and reactivate AChE. Atropine can be used as an antidote in conjunction with pralidoxime or other pyridinium oximes (such as trimedoxime or obidoxime), though the use of '-oximes' has been found to be of no benefit, or possibly harmful, in at least two meta-analyses. Atropine is a muscarinic antagonist, and thus blocks the action of acetylcholine peripherally. |
|---|
| Normal Concentrations |
|---|
| Not Available |
|---|
| Abnormal Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB01355 |
|---|
| PubChem Compound ID | 991 |
|---|
| ChEMBL ID | CHEMBL261919 |
|---|
| ChemSpider ID | 13844817 |
|---|
| KEGG ID | C06604 |
|---|
| UniProt ID | Not Available |
|---|
| OMIM ID | |
|---|
| ChEBI ID | 27928 |
|---|
| BioCyc ID | PARATHION |
|---|
| CTD ID | D010278 |
|---|
| Stitch ID | Parathion |
|---|
| PDB ID | Not Available |
|---|
| ACToR ID | 1126 |
|---|
| Wikipedia Link | Parathion |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Link |
|---|
| General References | - Mutch E, Daly AK, Leathart JB, Blain PG, Williams FM: Do multiple cytochrome P450 isoforms contribute to parathion metabolism in man? Arch Toxicol. 2003 Jun;77(6):313-20. Epub 2003 Mar 21. [12669189 ]
- Costa LG, Richter RJ, Li WF, Cole T, Guizzetti M, Furlong CE: Paraoxonase (PON 1) as a biomarker of susceptibility for organophosphate toxicity. Biomarkers. 2003 Jan-Feb;8(1):1-12. [12519632 ]
- Abel EL, Bammler TK, Eaton DL: Biotransformation of methyl parathion by glutathione S-transferases. Toxicol Sci. 2004 Jun;79(2):224-32. Epub 2004 Apr 21. [15103050 ]
- Wolf G: Serum retinol-binding protein: a link between obesity, insulin resistance, and type 2 diabetes. Nutr Rev. 2007 May;65(5):251-6. [17566551 ]
- Bohn G, Rucker G, Luckas KH: [Mass spectrometric and gaschromatographic detection of parathione in autopsy material after murder by poisoning]. Z Rechtsmed. 1971;68(1):45-52. [5547745 ]
- SALAMA S, EL-SHIHY A, IBRAHIM HH, KODSY A: THE ACTION OF PARATHIONE ON THE C.N.S. J Egypt Med Assoc. 1963;46:500-8. [14066548 ]
- Youssef SH, el-Sayed MG, Atef M: Influence of gentamicin and rifamycin on toxicity and biotransformation of methyl parathione in rats. Dtsch Tierarztl Wochenschr. 1987 Apr 8;94(4):203-5. [3297603 ]
- Innocenti A, Supuran CT: Paraoxon, 4-nitrophenyl phosphate and acetate are substrates of alpha- but not of beta-, gamma- and zeta-carbonic anhydrases. Bioorg Med Chem Lett. 2010 Nov 1;20(21):6208-12. doi: 10.1016/j.bmcl.2010.08.110. Epub 2010 Aug 26. [20833546 ]
- Kawahara K, Tanaka A, Yoon J, Yokota A: Reclassification of a parathione-degrading Flavobacterium sp. ATCC 27551 as Sphingobium fuliginis. J Gen Appl Microbiol. 2010 Jun;56(3):249-55. [20647682 ]
- Lotz W, Fasske E: [Pneumonitis with fatal pulmonary fibrosis (Hamman-Rich syndrome) due to parathion-(E-605-) poisoning]. Rofo. 1986 May;144(5):536-41. [3012677 ]
- SALAMA S, EL-SHISHY A, IBRAHIM HH: THE ACTION OF PARATHIONE ON THE CIRCULATORY & RESPIRATORY SYSTEMS. J Egypt Med Assoc. 1963;46:240-62. [14065272 ]
- Mueller RF, Hornung S, Furlong CE, Anderson J, Giblett ER, Motulsky AG: Plasma paraoxonase polymorphism: a new enzyme assay, population, family, biochemical, and linkage studies. Am J Hum Genet. 1983 May;35(3):393-408. [6305189 ]
- Lewis RJ (1996). Sax's Dangerous Properties of Industrial Materials. 9th ed. Volumes 1-3. New York, NY: Van Nostrand Reinhold.
- National Institute for Occupational Safety and Health (2002). RTECS: Registry of Toxic Effects of Chemical Substances.
- NIOSH: The National Institute for Occupational Safety and Health (1976). Criteria Document: Parathion.
- International Agency for Research on Cancer (2014). IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. [Link]
- Wikipedia. Parathion. Last Updated 9 June 2009. [Link]
- ATSDR - Agency for Toxic Substances and Disease Registry (2001). Toxicological profile for methyl parathion U.S. Public Health Service in collaboration with U.S. Environmental Protection Agency (EPA). [Link]
|
|---|
| Gene Regulation |
|---|
| Up-Regulated Genes | | Gene | Gene Symbol | Gene ID | Interaction | Chromosome | Details |
|---|
|
|---|
| Down-Regulated Genes | | Gene | Gene Symbol | Gene ID | Interaction | Chromosome | Details |
|---|
|
|---|