| Record Information |
|---|
| Version | 2.0 |
|---|
| Creation Date | 2009-03-06 18:58:41 UTC |
|---|
| Update Date | 2014-12-24 20:21:46 UTC |
|---|
| Accession Number | T3D0389 |
|---|
| Identification |
|---|
| Common Name | Biphenyl |
|---|
| Class | Small Molecule |
|---|
| Description | Biphenyl is found in alcoholic beverages. Fungistat, especially for citrus fruits. Biphenyl is used as food preservative and flavouring agent. Biphenyl is detected in bilberry, wine grape, carrot, peas, rum, potato, bell pepper, tomato, butter, milk, smoked fatty fish, cocoa, coffee, roast peanuts, olive, buckwheat and tamarind. Generally, the fruit packaging is impregnated with biphenyl, which evaporates into the air space surrounding the fruit. Some biphenyl is absorbed by the fruit skins
Biphenyl has been shown to exhibit anti-coagulant and catabolic functions (5, 6).
Biphenyl belongs to the family of Biphenyls and Derivatives. These are organic compounds containing to benzene rings linked together by a C-C bond. |
|---|
| Compound Type | - Aromatic Hydrocarbon
- Coolant
- Food Toxin
- Industrial/Workplace Toxin
- Metabolite
- Natural Compound
- Organic Compound
- Organochloride
- Plasticizer
- Pollutant
- Polychlorinated Biphenyl
|
|---|
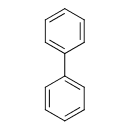
| Chemical Structure | |
|---|
| Synonyms | | Synonym | | 1, 1'-Diphenyl | | 1,1'-Biphenyl | | Bibenzene | | Diphenyl | | FEMA 3129 | | Lemonene | | Phenador-X | | Phenylbenzene | | PHPH | | Tetrosin LY | | Xenene |
|
|---|
| Chemical Formula | C12H10 |
|---|
| Average Molecular Mass | 154.208 g/mol |
|---|
| Monoisotopic Mass | 154.078 g/mol |
|---|
| CAS Registry Number | 92-52-4 |
|---|
| IUPAC Name | 1,1'-biphenyl |
|---|
| Traditional Name | diphenyl |
|---|
| SMILES | C1=CC=C(C=C1)C1=CC=CC=C1 |
|---|
| InChI Identifier | InChI=1S/C12H10/c1-3-7-11(8-4-1)12-9-5-2-6-10-12/h1-10H |
|---|
| InChI Key | InChIKey=ZUOUZKKEUPVFJK-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as biphenyls and derivatives. These are organic compounds containing to benzene rings linked together by a C-C bond. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Benzene and substituted derivatives |
|---|
| Sub Class | Biphenyls and derivatives |
|---|
| Direct Parent | Biphenyls and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Biphenyl
- Aromatic hydrocarbon
- Unsaturated hydrocarbon
- Hydrocarbon
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Exogenous |
|---|
| Cellular Locations | |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | |
|---|
| Biological Roles | |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Appearance | White scales (2). |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | 71°C | | Boiling Point | 254.5°C (490.1°F) | | Solubility | 0.00748 mg/mL at 25°C | | LogP | 4.01 |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | Deposition Date | View |
|---|
| GC-MS | GC-MS Spectrum - EI-B (Non-derivatized) | splash10-0udi-8900000000-e5b4f289ac40ae7f058d | 2017-09-12 | View Spectrum | | GC-MS | GC-MS Spectrum - EI-B (Non-derivatized) | splash10-0udi-3900000000-11fa850b6e97cb680edd | 2017-09-12 | View Spectrum | | GC-MS | GC-MS Spectrum - EI-B (Non-derivatized) | splash10-0udi-2900000000-59a470228d72016ea76f | 2017-09-12 | View Spectrum | | GC-MS | GC-MS Spectrum - EI-B (Non-derivatized) | splash10-0udi-0900000000-01b08aa1cb97e4f8749a | 2017-09-12 | View Spectrum | | GC-MS | GC-MS Spectrum - EI-B (Non-derivatized) | splash10-0udi-1900000000-4ed186df67a09ce0650f | 2017-09-12 | View Spectrum | | GC-MS | GC-MS Spectrum - EI-B (Non-derivatized) | splash10-0udi-3900000000-b083707713188a3b56af | 2017-09-12 | View Spectrum | | GC-MS | GC-MS Spectrum - EI-B (Non-derivatized) | splash10-0udi-2900000000-931304319dd9f132e118 | 2017-09-12 | View Spectrum | | GC-MS | GC-MS Spectrum - EI-B (Non-derivatized) | splash10-0udi-6900000000-f9f4bb759b70237cae7b | 2017-09-12 | View Spectrum | | GC-MS | GC-MS Spectrum - CI-B (Non-derivatized) | splash10-0a4i-0900000000-5043f3793673c0f94d67 | 2017-09-12 | View Spectrum | | GC-MS | GC-MS Spectrum - GC-EI-TOF (Non-derivatized) | splash10-0udi-0900000000-77f5c79fcaa35c93a5b8 | 2017-09-12 | View Spectrum | | GC-MS | GC-MS Spectrum - EI-B (Non-derivatized) | splash10-0udi-8900000000-e5b4f289ac40ae7f058d | 2018-05-18 | View Spectrum | | GC-MS | GC-MS Spectrum - EI-B (Non-derivatized) | splash10-0udi-3900000000-11fa850b6e97cb680edd | 2018-05-18 | View Spectrum | | GC-MS | GC-MS Spectrum - EI-B (Non-derivatized) | splash10-0udi-2900000000-59a470228d72016ea76f | 2018-05-18 | View Spectrum | | GC-MS | GC-MS Spectrum - EI-B (Non-derivatized) | splash10-0udi-0900000000-01b08aa1cb97e4f8749a | 2018-05-18 | View Spectrum | | GC-MS | GC-MS Spectrum - EI-B (Non-derivatized) | splash10-0udi-1900000000-4ed186df67a09ce0650f | 2018-05-18 | View Spectrum | | GC-MS | GC-MS Spectrum - EI-B (Non-derivatized) | splash10-0udi-3900000000-b083707713188a3b56af | 2018-05-18 | View Spectrum | | GC-MS | GC-MS Spectrum - EI-B (Non-derivatized) | splash10-0udi-2900000000-931304319dd9f132e118 | 2018-05-18 | View Spectrum | | GC-MS | GC-MS Spectrum - EI-B (Non-derivatized) | splash10-0udi-6900000000-f9f4bb759b70237cae7b | 2018-05-18 | View Spectrum | | GC-MS | GC-MS Spectrum - CI-B (Non-derivatized) | splash10-0a4i-0900000000-5043f3793673c0f94d67 | 2018-05-18 | View Spectrum | | GC-MS | GC-MS Spectrum - GC-EI-TOF (Non-derivatized) | splash10-0udi-0900000000-77f5c79fcaa35c93a5b8 | 2018-05-18 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0udi-1900000000-56e5bf47be0405d6836b | 2016-09-22 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0900000000-edde0dcdd8d1f5866932 | 2016-06-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4i-0900000000-e45b080cc4494ae7ace3 | 2016-06-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0pdr-3900000000-eeea5f59ba3188214b77 | 2016-06-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0900000000-f405b638fff04b7f3ea6 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-0900000000-f405b638fff04b7f3ea6 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0udi-0900000000-5347e97776fd82473263 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0900000000-4be39230312831d721f7 | 2021-09-22 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4i-0900000000-49244566d5b6e3ac17e6 | 2021-09-22 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0ufr-0900000000-d41e8809ac37a183033c | 2021-09-22 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0900000000-8b214a52c8e78f3210f4 | 2021-09-22 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-0900000000-8b214a52c8e78f3210f4 | 2021-09-22 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0udi-0900000000-43cb10c15248f4da21d0 | 2021-09-22 | View Spectrum | | MS | Mass Spectrum (Electron Ionization) | splash10-0udi-3900000000-92b73f1f35424a0fd73d | 2014-09-20 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, 400 MHz, CDCl3, experimental) | Not Available | 2014-09-20 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, 100.41 MHz, CDCl3, experimental) | Not Available | 2014-09-23 | View Spectrum | | 1D NMR | 1H NMR Spectrum (1D, CDCl3, experimental) | Not Available | 2016-10-22 | View Spectrum | | 1D NMR | 13C NMR Spectrum (1D, CDCl3, experimental) | Not Available | 2016-10-22 | View Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Inhalation (3) ; dermal (3) ; eye contact (3) |
|---|
| Mechanism of Toxicity | Biphenyl alters the permeability properties of mitochondrial membranes (1). |
|---|
| Metabolism | Diphenyl is well absorbed through the gastrointestinal tract. It is rapidly metabolized to 4-hydroxybiphenyl, 4-phenyl-catechol and 4,4'-dihydroxyphenyl, which are excreted in urine and bile as glucuronide and mercapturic conjugates. The metabolites of diphenyl, mainly 4-hydroxybiphenyl, are excreted rapidly and almost exclusively in the urine. Acute oral toxicity is moderate. (9, 4). |
|---|
| Toxicity Values | LD50: 3280 mg/kg (Oral, Rat) (11)
LD50: 2400 mg/kg (Oral, Rabbit) (7)
LD50: 2500 mg/kg (Dermal, Rabbit) (10) |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | No indication of carcinogenicity (not listed by IARC). (17) |
|---|
| Uses/Sources | Organic synthesis; heat transfer agent; fungistat in packaging of citrus fruit; plant disease control; manufacture of benzidine; dyeing assistant for polyesters (2). |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Exposure to diphenyl can cause necrosis in the liver and kidney, with regions of cirrhosis in liver; also, degenerative changes in heart muscle, damage to the central and peripheral nervous systems, as well as death can result from diphenyl poisoning (9). |
|---|
| Symptoms | Symptoms of diphenyl poisoning include headache, fatigue, abdominal pain with nausea or diarrhea and various symptoms, vomiting and bronchitis (9). |
|---|
| Treatment | Consider gastric lavage, after ingestion of a potentially life-threatening amount of poison if it can be performed soon after ingestion. In case of inhalation exposure, move patient to fresh air and monitor for respiratory distress. If cough or difficulty breathing develops, evaluate for respiratory tract irritation, bronchitis, or pneumonitis. Administer oxygen and assist ventilation as required. Treat bronchospasm with inhaled beta2 agonist and oral or parenteral corticosteroids. In case of eye exposure, irrigate exposed eyes with copious amounts of room temperature water for at least 15 minutes. I f the exposure occurred through dermal contact, remove contaminated clothing and wash exposed area thoroughly with soap and water. (8)
|
|---|
| Normal Concentrations |
|---|
| Not Available |
|---|
| Abnormal Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB34437 |
|---|
| PubChem Compound ID | 7095 |
|---|
| ChEMBL ID | CHEMBL14092 |
|---|
| ChemSpider ID | 6828 |
|---|
| KEGG ID | C06588 |
|---|
| UniProt ID | Not Available |
|---|
| OMIM ID | |
|---|
| ChEBI ID | 17097 |
|---|
| BioCyc ID | CPD-945 |
|---|
| CTD ID | C010574 |
|---|
| Stitch ID | Diphenyl |
|---|
| PDB ID | BNL |
|---|
| ACToR ID | 162 |
|---|
| Wikipedia Link | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Link |
|---|
| General References | - Nishihara Y: Comparative study of the effects of biphenyl and Kanechlor-400 on the respiratory and energy linked activities of rat liver mitochondria. Br J Ind Med. 1985 Feb;42(2):128-32. [3918562 ]
- Maeda A, Maeda T, Imanishi Y, Golczak M, Moise AR, Palczewski K: Aberrant metabolites in mouse models of congenital blinding diseases: formation and storage of retinyl esters. Biochemistry. 2006 Apr 4;45(13):4210-9. [16566595 ]
- Wigg SJ, Tare M, Forbes J, Cooper ME, Thomas MC, Coleman HA, Parkington HC, O'Brien RC: Early vitamin E supplementation attenuates diabetes-associated vascular dysfunction and the rise in protein kinase C-beta in mesenteric artery and ameliorates wall stiffness in femoral artery of Wistar rats. Diabetologia. 2004 Jun;47(6):1038-46. Epub 2004 Jun 8. [15184978 ]
- Huang Y, Ishizuka T, Miura A, Kajita K, Ishizawa M, Kimura M, Yamamoto Y, Kawai Y, Morita H, Uno Y, Yasuda K: Effect of 1 alpha,25-dihydroxy vitamin D3 and vitamin E on insulin-induced glucose uptake in rat adipocytes. Diabetes Res Clin Pract. 2002 Mar;55(3):175-83. [11850093 ]
- Jia ZJ, Wu Y, Huang W, Zhang P, Clizbe LA, Goldman EA, Sinha U, Arfsten AE, Edwards ST, Alphonso M, Hutchaleelaha A, Scarborough RM, Zhu BY: 1-(2-Naphthyl)-1H-pyrazole-5-carboxylamides as potent factor Xa inhibitors. Part 2: A survey of P4 motifs. Bioorg Med Chem Lett. 2004 Mar 8;14(5):1221-7. [14980670 ]
- Kosono S, Maeda M, Fuji F, Arai H, Kudo T: Three of the seven bphC genes of Rhodococcus erythropolis TA421, isolated from a termite ecosystem, are located on an indigenous plasmid associated with biphenyl degradation. Appl Environ Microbiol. 1997 Aug;63(8):3282-5. [9251216 ]
- American Conference of Governmental Industrial Hygienists (2001). Documentation of Threshold Limit Values for Chemical Substances and Physical Agents and Biological Exposure Indices for 2001. Cincinnati, OH: American Conference of Governmental Industrial Hygienists.
- Rumack BH (2009). POISINDEX(R) Information System. Englewood, CO: Micromedex, Inc. CCIS Volume 141, edition expires Aug, 2009.
- Gosselin RE, Smith RP, and Hodge HC (1984). Clinical Toxicology of Commercial Products. 5th ed. Baltimore: Williams and Wilkins.
- Kirk-Othmer Encyclopedia of Chemical Technology (1978-1984). 3rd ed. Volumes 1-26. New York, NY: John Wiley and Sons.
- O'Neil MJ (ed) (2001). The Merck Index - An Encyclopedia of Chemicals, Drugs, and Biologicals. 13th ed. Whitehouse Station, NJ: Merck and Co., Inc.
- Lewis RJ Sr. (ed) (1997). Hawley's Condensed Chemical Dictionary. 13th ed. New York, NY: Van Nostrand Rheinhold Co.
- NIOSH: The National Institute for Occupational Safety and Health (1983). National Occupational Exposure Survey (NOES).
- World Health Organization/International Programme on Chemical Safety (1999). Concise International Chemical Assessment Document No. 6. Biphenyl.
- Yannai, Shmuel. (2004) Dictionary of food compounds with CD-ROM: Additives, flavors, and ingredients. Boca Raton: Chapman & Hall/CRC.
- ATSDR - Agency for Toxic Substances and Disease Registry (2000). Toxicological profile for polychlorinated biphenyls. U.S. Public Health Service in collaboration with U.S. Environmental Protection Agency (EPA). [Link]
- International Agency for Research on Cancer (2014). IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. [Link]
|
|---|
| Gene Regulation |
|---|
| Up-Regulated Genes | Not Available |
|---|
| Down-Regulated Genes | Not Available |
|---|