
Browsing Toxins
| T3DB ID | Name CAS Number | Formula Weight | Structure | Type | Mechanism of Toxicity |
|---|---|---|---|---|---|
| T3D2467 | Phalloidin 17466-45-4 | Not Available 3301.700 g/mol |  |
| It binds actin, preventing its depolymerization and poisoning the cell. Phalloidin binds specifically at the interface between F-actin subunits, locking adjacent subun...more Number of Targets: 6 |
| T3D0362 | Mercuric potassium iodide 7783-33-7 | HgI4K2 786.400 g/mol | 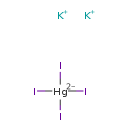 |
| High-affinity binding of the divalent mercuric ion to thiol or sulfhydryl groups of proteins is believed to be the major mechanism for the activity of mercury. Through...more Number of Targets: 50 |
| T3D1993 | 2,2',3,3',4,5',6,6'-Octabromobiphenyl 69887-11-2 | C12H2Br8 785.376 g/mol | 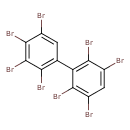 |
| The exact mechanism of toxicty of PBBs varies depending on the specific congener. The predominant interaction is believed to involve the aryl hydrocarbon receptor (AhR...more Number of Targets: 1 |
| T3D1991 | 2,2',3,3',4,4',5',6-Octabromobiphenyl 69278-61-1 | C12H2Br8 785.376 g/mol | 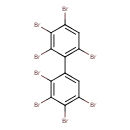 |
| The exact mechanism of toxicty of PBBs varies depending on the specific congener. The predominant interaction is believed to involve the aryl hydrocarbon receptor (AhR...more Number of Targets: 1 |
| T3D1988 | 2,2',3,3',4,4',5,5'-Octabromobiphenyl 67889-00-3 | C12H2Br8 785.376 g/mol | 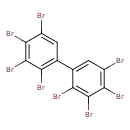 |
| The exact mechanism of toxicty of PBBs varies depending on the specific congener. The predominant interaction is believed to involve the aryl hydrocarbon receptor (AhR...more Number of Targets: 1 |
| T3D1971 | 2,2',3,3',5,5',6,6'-Octabromobiphenyl 59080-41-0 | C12H2Br8 785.376 g/mol | 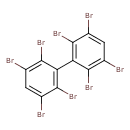 |
| The exact mechanism of toxicty of PBBs varies depending on the specific congener. The predominant interaction is believed to involve the aryl hydrocarbon receptor (AhR...more Number of Targets: 1 |
| T3D1948 | Octabromobiphenyl 27858-07-7 | C12H2Br8 785.376 g/mol | 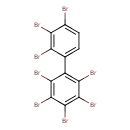 |
| The exact mechanism of toxicty of PBBs varies depending on the specific congener. The predominant interaction is believed to involve the aryl hydrocarbon receptor (AhR...more Number of Targets: 1 |
| T3D1934 | 2,2',3,4,4',5,6,6'-Octabromobiphenyl 119264-61-8 | C12H2Br8 785.376 g/mol | 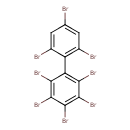 |
| The exact mechanism of toxicty of PBBs varies depending on the specific congener. The predominant interaction is believed to involve the aryl hydrocarbon receptor (AhR...more Number of Targets: 1 |
| T3D1933 | 2,2',3,3',4,5,6,6'-Octabromobiphenyl 119264-60-7 | C12H2Br8 785.376 g/mol | 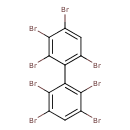 |
| The exact mechanism of toxicty of PBBs varies depending on the specific congener. The predominant interaction is believed to involve the aryl hydrocarbon receptor (AhR...more Number of Targets: 1 |
| T3D1932 | 2,2',3,3',4,4',6,6'-Octabromobiphenyl 119264-59-4 | C12H2Br8 785.376 g/mol | 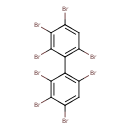 |
| The exact mechanism of toxicty of PBBs varies depending on the specific congener. The predominant interaction is believed to involve the aryl hydrocarbon receptor (AhR...more Number of Targets: 1 |
| T3D3740 | Beauvericin 26048-05-5 | C45H57N3O9 783.949 g/mol | 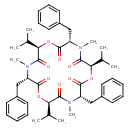 |
| Beauvericin is cytotoxic and increases ion permeability in cell membranes by forming complexes with essential cations (Ca2+, Na+, K+) and by forming cation-selective c...more Number of Targets: 3 |
| T3D2670 | Digoxin 20830-75-5 | C41H64O14 780.939 g/mol | 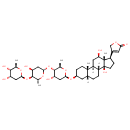 |
| Digoxin binds to a site on the extracellular aspect of the alpha-subunit of the Na+/K+ ATPase pump in the membranes of heart cells (myocytes) and decreases its functio...more Number of Targets: 17 |
| T3D1330 | Basic lead carbonate 1319-46-6 | C2H2O8Pb3 775.600 g/mol | 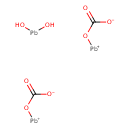 |
| Lead mimics other biologically important metals, such as zinc, calcium, and iron, competing as cofactors for many of their respective enzymatic reactions. For example,...more Number of Targets: 29 |
| T3D1324 | Lead stearate 1072-35-1 | C36H70O4Pb 774.100 g/mol |  |
| Lead mimics other biologically important metals, such as zinc, calcium, and iron, competing as cofactors for many of their respective enzymatic reactions. For example,...more Number of Targets: 29 |
| T3D2469 | Phalloin 28227-92-1 | C35H48N8O10S 772.868 g/mol | 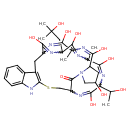 |
| Phallotoxins bind actin, preventing its depolymerization and poisoning the cell. They bind specifically at the interface between F-actin subunits, locking adjacent sub...more Number of Targets: 6 |
| T3D4021 | Digitoxin 71-63-6 | C41H64O13 764.939 g/mol | 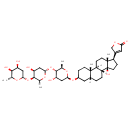 |
| Digitoxin inhibits the Na-K-ATPase membrane pump, resulting in an increase in intracellular sodium and calcium concentrations. Increased intracellular concentrations o...more Number of Targets: 2 |
| T3D4916 | Fast green FCF 2353-45-9 | C37H36N2O10S3 764.884 g/mol | 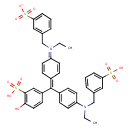 |
| Not Available Number of Targets: 5 |
| T3D1349 | Mercuric oleate 1191-80-6 | C36H66HgO4 763.500 g/mol |  |
| High-affinity binding of the divalent mercuric ion to thiol or sulfhydryl groups of proteins is believed to be the major mechanism for the activity of mercury. Through...more Number of Targets: 50 |
| T3D1137 | Mangafodipir 140678-14-4 | C22H27MnN4Na3O14P2 757.323 g/mol | 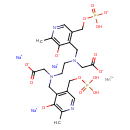 |
| Manganese is a cellular toxicant that can impair transport systems, enzyme activities, and receptor functions. It primarily targets the central nervous system, particu...more Number of Targets: 5 |
| T3D2468 | Prophalloin 67739-84-8 | C35H48N8O9S 756.869 g/mol | 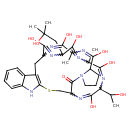 |
| Phallotoxins bind actin, preventing its depolymerization and poisoning the cell. They bind specifically at the interface between F-actin subunits, locking adjacent sub...more Number of Targets: 6 |
| T3D2479 | Vindesine 53643-48-4 | C43H55N5O7 753.926 g/mol | 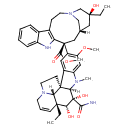 |
| Vindesine acts by causing the arrest of cells in metaphase mitosis through its inhibition tubulin mitotic funcitoning. The drug is cell-cycle specific for the S phase. Number of Targets: 1 |
| T3D1361 | Merbromin 129-16-8 | C20H8Br2HgNa2O6 750.650 g/mol | 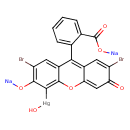 |
| High-affinity binding of the divalent mercuric ion to thiol or sulfhydryl groups of proteins is believed to be the major mechanism for the activity of mercury. Through...more Number of Targets: 56 |
| T3D1280 | Monomethyltin tris(isooctyl mercaptoacetate) 54849-38-6 | C31H60O6S3Sn 743.710 g/mol | 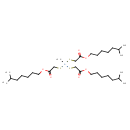 |
| Organotin compounds produce neurotoxic and immunotoxic effects. Organotins may directly activate glial cells contributing to neuronal cell degeneration by local releas...more Number of Targets: 12 |
| T3D4764 | Erythromycin 114-07-8 | C37H67NO13 733.927 g/mol |  |
| Erythromycin acts by penetrating the bacterial cell membrane and reversibly binding to the 50 S subunit of bacterial ribosomes or near the “P” or donor site so that bi...more Number of Targets: 1 |
| T3D1345 | Mercury oxide sulfate 1312-03-4 | Hg3O6S 729.830 g/mol |  |
| High-affinity binding of the divalent mercuric ion to thiol or sulfhydryl groups of proteins is believed to be the major mechanism for the activity of mercury. Through...more Number of Targets: 50 |