| Record Information |
|---|
| Version | 2.0 |
|---|
| Creation Date | 2009-07-30 17:58:51 UTC |
|---|
| Update Date | 2014-12-24 20:26:07 UTC |
|---|
| Accession Number | T3D3501 |
|---|
| Identification |
|---|
| Common Name | Docetaxel |
|---|
| Class | Small Molecule |
|---|
| Description | Docetaxel is a clinically well established anti-mitotic chemotherapy medication used mainly for the treatment of breast, ovarian, and non-small cell lung cancer. Docetaxel binds to microtubules reversibly with high affinity and has a maximum stoichiometry of one mole docetaxel per mole tubulin in microtubules. |
|---|
| Compound Type | - Amine
- Antimalarial
- Antineoplastic Agent
- Antineoplastic Agent, Phytogenic
- Drug
- Ester
- Ether
- Metabolite
- Organic Compound
- Radiation-Sensitizing Agent
- Synthetic Compound
|
|---|
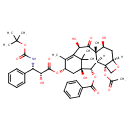
| Chemical Structure | |
|---|
| Synonyms | | Synonym | | Docefrez | | Docetaxel anhydrous | | Docetaxel, Trihydrate | | N-Debenzoyl-N-(tert-butoxycarbonyl)-10-deacetylpaclitaxel | | N-Debenzoyl-N-(tert-butoxycarbonyl)-10-deacetyltaxol | | Taxotere | | TXL |
|
|---|
| Chemical Formula | C43H53NO14 |
|---|
| Average Molecular Mass | 807.879 g/mol |
|---|
| Monoisotopic Mass | 807.347 g/mol |
|---|
| CAS Registry Number | 114977-28-5 |
|---|
| IUPAC Name | (1S,2S,3R,4S,7R,9S,10S,12R,15S)-4-(acetyloxy)-15-{[(2R,3S)-3-{[(tert-butoxy)carbonyl]amino}-2-hydroxy-3-phenylpropanoyl]oxy}-1,9,12-trihydroxy-10,14,17,17-tetramethyl-11-oxo-6-oxatetracyclo[11.3.1.0^{3,10}.0^{4,7}]heptadec-13-en-2-yl benzoate |
|---|
| Traditional Name | (1S,2S,3R,4S,7R,9S,10S,12R,15S)-4-(acetyloxy)-15-{[(2R,3S)-3-[(tert-butoxycarbonyl)amino]-2-hydroxy-3-phenylpropanoyl]oxy}-1,9,12-trihydroxy-10,14,17,17-tetramethyl-11-oxo-6-oxatetracyclo[11.3.1.0^{3,10}.0^{4,7}]heptadec-13-en-2-yl benzoate |
|---|
| SMILES | [H][C@](O)(C(=O)O[C@@]1([H])C[C@@]2(O)[C@@]([H])(OC(=O)C3=CC=CC=C3)[C@]3([H])[C@@]4(CO[C@]4([H])C[C@]([H])(O)[C@@]3(C)C(=O)[C@]([H])(O)C(=C1C)C2(C)C)OC(C)=O)[C@@]([H])(N=C(O)OC(C)(C)C)C1=CC=CC=C1 |
|---|
| InChI Identifier | InChI=1S/C43H53NO14/c1-22-26(55-37(51)32(48)30(24-15-11-9-12-16-24)44-38(52)58-39(3,4)5)20-43(53)35(56-36(50)25-17-13-10-14-18-25)33-41(8,34(49)31(47)29(22)40(43,6)7)27(46)19-28-42(33,21-54-28)57-23(2)45/h9-18,26-28,30-33,35,46-48,53H,19-21H2,1-8H3,(H,44,52)/t26-,27-,28+,30-,31+,32+,33-,35-,41+,42-,43+/m0/s1 |
|---|
| InChI Key | InChIKey=ZDZOTLJHXYCWBA-VCVYQWHSSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as taxanes and derivatives. These are diterpenoids with a structure based either on the taxane skeleton, or a derivative thereof. In term of phytochemistry, several derivatives of the taxane skeleton exist: 2(3->20)-abeotaxane, 3,11-cyclotaxane, 11(15->1),11(10->9)-abeotaxane, 3,8-seco-taxane, and 11(15->1)-abeotaxane, among others. More complex skeletons have been found recently, which include the taxane-derived [3.3.3] propellane ring system. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Diterpenoids |
|---|
| Direct Parent | Taxanes and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Taxane diterpenoid
- Benzoate ester
- Benzoic acid or derivatives
- Tricarboxylic acid or derivatives
- Benzoyl
- Fatty acid ester
- Monocyclic benzene moiety
- Fatty acyl
- Monosaccharide
- Benzenoid
- Cyclic alcohol
- Carbamic acid ester
- Tertiary alcohol
- Carboxylic acid ester
- Ketone
- Carbonic acid derivative
- Oxetane
- Secondary alcohol
- Organoheterocyclic compound
- Oxacycle
- Ether
- Dialkyl ether
- Carboxylic acid derivative
- Polyol
- Organic nitrogen compound
- Hydrocarbon derivative
- Alcohol
- Carbonyl group
- Organic oxide
- Organonitrogen compound
- Organooxygen compound
- Organic oxygen compound
- Organopnictogen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Exogenous |
|---|
| Cellular Locations | - Cytoplasm
- Extracellular
- Membrane
|
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | |
|---|
| Pathways | | Name | SMPDB Link | KEGG Link |
|---|
| Docetaxel Pathway | Not Available | Not Available |
|
|---|
| Applications | |
|---|
| Biological Roles | |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Appearance | White powder. |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | 232°C | | Boiling Point | Not Available | | Solubility | Insoluble | | LogP | 2.4 |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | Deposition Date | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_1) - 70eV, Positive | Not Available | 2021-10-19 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_2) - 70eV, Positive | Not Available | 2021-10-19 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_3) - 70eV, Positive | Not Available | 2021-10-19 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_4) - 70eV, Positive | Not Available | 2021-10-19 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_5) - 70eV, Positive | Not Available | 2021-10-19 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_6) - 70eV, Positive | Not Available | 2021-10-19 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 35V, Positive | splash10-056r-0597250110-87ceab2cb1907efad869 | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 35V, Negative | splash10-0kmi-0069066000-3564c38332034a6ece44 | 2021-09-20 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0ke9-2110021900-f5d0bc483676fe5dc3e1 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-056r-6520040900-5dc2943e6be89bc86b0c | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a6r-6900660000-2867151287c5a0346e02 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-05iu-4410040920-b828ec76844ebea2d7a8 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-05i3-9210270300-f8577941b1621d9b82e5 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0ab9-9300140000-7ac14648b028058fac74 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-003r-0390210310-072c630f13bc0f81269a | 2021-10-11 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-004i-1590201200-973eb03969da7056a8a6 | 2021-10-11 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0abc-3910101210-42478cd96d93fb415eeb | 2021-10-11 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-3390000110-65c0d7adc2d422e11f9c | 2021-10-11 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0pdi-5940101000-da55ca527881bac1ac24 | 2021-10-11 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0059-9601000000-3666f54ffa6e69c8958a | 2021-10-11 | View Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Intravenous injection. The pharmacokinetic profile is consistent with a three-compartment model. The area under the curve (AUC) was dose proportional following doses of 70 mg/m2 to 115 mg/m2 with infusion times of 1 to 2 hours. |
|---|
| Mechanism of Toxicity | Docetaxel interferes with the normal function of microtubule growth. Whereas drugs like colchicine cause the depolymerization of microtubules in vivo, docetaxel arrests their function by having the opposite effect; it hyper-stabilizes their structure. This destroys the cell's ability to use its cytoskeleton in a flexible manner. Specifically, docetaxel binds to the β-subunit of tubulin. Tubulin is the 'building block' of mictotubules, and the binding of docetaxel locks these building blocks in place. The resulting microtubule/docetaxel complex does not have the ability to disassemble. This adversely affects cell function because the shortening and lengthening of microtubules (termed dynamic instability) is necessary for their function as a transportation highway for the cell. Chromosomes, for example, rely upon this property of microtubules during mitosis. Further research has indicated that docetaxel induces programmed cell death (apoptosis) in cancer cells by binding to an apoptosis stopping protein called Bcl-2 (B-cell leukemia 2) and thus arresting its function. |
|---|
| Metabolism | Hepatic. In vitro drug interaction studies revealed that docetaxel is metabolized by the CYP3A4 isoenzyme (1 major, 3 minor metabolites).
Route of Elimination: Docetaxel was eliminated in both the urine and feces following oxidative metabolism of the tert-butyl ester group, but fecal excretion was the main elimination route. Within 7 days, urinary and fecal excretion accounted for approximately 6% and 75% of the administered radioactivity, respectively.
Half Life: Dose-dependent. Doses of 70 mg per square meter of body surface area (mg/m 2 ) or higher produce a triphasic elimination profile. With lower doses, assay limitations precluded detection of the terminal elimination phase. The half-life of the alpha, beta, and gamma phase are 4 minutes, 36 minutes, and 11.1 hours, respectively. |
|---|
| Toxicity Values | Oral LD50 in rat is >2000 mg/kg. |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | No indication of carcinogenicity to humans (not listed by IARC). |
|---|
| Uses/Sources | For the treatment of patients with locally advanced or metastatic breast cancer after failure of prior chemotherapy. Also used as a single agent in the treatment of patients with locally advanced or metastatic non-small cell lung cancer after failure of prior platinum-based chemotherapy. It is also used in combination with prednisone, in the treatment of patients with androgen independent (hormone refractory) metastatic prostate cancer. Furthermore, docetaxel has uses in the treatment of gastric adenocarinoma and head and neck cancer. |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Anticipated complications of overdosage include: bone marrow suppression, peripheral neurotoxicity, and mucositis. In two reports of overdose, one patient received 150 mg/m2 and the other received 200 mg/m2 as 1-hour infusions. Both patients experienced severe neutropenia, mild asthenia, cutaneous reactions, and mild paresthesia, and recovered without incident. |
|---|
| Treatment | Not Available |
|---|
| Normal Concentrations |
|---|
| Not Available |
|---|
| Abnormal Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB01248 |
|---|
| HMDB ID | HMDB15378 |
|---|
| PubChem Compound ID | 148124 |
|---|
| ChEMBL ID | CHEMBL92 |
|---|
| ChemSpider ID | 130581 |
|---|
| KEGG ID | C11231 |
|---|
| UniProt ID | Not Available |
|---|
| OMIM ID | |
|---|
| ChEBI ID | 4672 |
|---|
| BioCyc ID | Not Available |
|---|
| CTD ID | Not Available |
|---|
| Stitch ID | Docetaxel |
|---|
| PDB ID | TXL |
|---|
| ACToR ID | Not Available |
|---|
| Wikipedia Link | Docetaxel |
|---|
| References |
|---|
| Synthesis Reference | Nicholas J. Sisti, Charles S. Swindell, “Method for docetaxel synthesis.” U.S. Patent US5688977, issued September, 1991. |
|---|
| MSDS | Link |
|---|
| General References | - FDA label
|
|---|
| Gene Regulation |
|---|
| Up-Regulated Genes | | Gene | Gene Symbol | Gene ID | Interaction | Chromosome | Details |
|---|
|
|---|
| Down-Regulated Genes | | Gene | Gene Symbol | Gene ID | Interaction | Chromosome | Details |
|---|
|
|---|