| Record Information |
|---|
| Version | 2.0 |
|---|
| Creation Date | 2010-05-20 15:08:50 UTC |
|---|
| Update Date | 2014-12-24 20:26:30 UTC |
|---|
| Accession Number | T3D3764 |
|---|
| Identification |
|---|
| Common Name | Janthitrem E |
|---|
| Class | Small Molecule |
|---|
| Description | Janthitrem E is produced by Penicillium janthinellum. Tremorgenic mycotoxin
Janthitrem e belongs to the family of Diterpenes. These are terpene compounds formed by four isoprene units. |
|---|
| Compound Type | - Ether
- Food Toxin
- Fungal Toxin
- Metabolite
- Mycotoxin
- Natural Compound
- Organic Compound
|
|---|
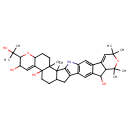
| Chemical Structure | |
|---|
| Synonyms | |
|---|
| Chemical Formula | C37H49NO6 |
|---|
| Average Molecular Mass | 603.788 g/mol |
|---|
| Monoisotopic Mass | 603.356 g/mol |
|---|
| CAS Registry Number | 90986-50-8 |
|---|
| IUPAC Name | 8-(2-hydroxypropan-2-yl)-2,3,23,23,25,25-hexamethyl-7,24-dioxa-31-azaoctacyclo[15.14.0.0²,¹⁵.0³,¹².0⁶,¹¹.0¹⁸,³⁰.0²⁰,²⁸.0²²,²⁷]hentriaconta-1(17),10,18(30),19,26,28-hexaene-9,12,21-triol |
|---|
| Traditional Name | 8-(2-hydroxypropan-2-yl)-2,3,23,23,25,25-hexamethyl-7,24-dioxa-31-azaoctacyclo[15.14.0.0²,¹⁵.0³,¹².0⁶,¹¹.0¹⁸,³⁰.0²⁰,²⁸.0²²,²⁷]hentriaconta-1(17),10,18(30),19,26,28-hexaene-9,12,21-triol |
|---|
| SMILES | CC(C)(O)C1OC2CCC3(C)C4(C)C(CC5=C4NC4=C5C=C5C(O)C6C(=CC(C)(C)OC6(C)C)C5=C4)CCC3(O)C2=CC1O |
|---|
| InChI Identifier | InChI=1/C37H49NO6/c1-32(2)17-23-19-15-25-20(14-21(19)29(40)28(23)34(5,6)44-32)22-13-18-9-12-37(42)24-16-26(39)31(33(3,4)41)43-27(24)10-11-35(37,7)36(18,8)30(22)38-25/h14-18,26-29,31,38-42H,9-13H2,1-8H3 |
|---|
| InChI Key | InChIKey=TVRIMSLYKUNOPS-UHFFFAOYNA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as naphthopyrans. Naphthopyrans are compounds containing a pyran ring fused to a naphthalene moiety. Furan is a 6 membered-ring non-aromatic ring with five carbon and one oxygen atoms. Naphthalene is a polycyclic aromatic hydrocarbon made up of two fused benzene rings. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Naphthopyrans |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Naphthopyrans |
|---|
| Alternative Parents | |
|---|
| Substituents | - Naphthopyran
- Naphthalene
- 3-alkylindole
- Indane
- Indole
- Indole or derivatives
- Pyran
- Benzenoid
- Cyclic alcohol
- Tertiary alcohol
- Pyrrole
- Heteroaromatic compound
- Secondary alcohol
- Azacycle
- Oxacycle
- Dialkyl ether
- Ether
- Polyol
- Organopnictogen compound
- Organic oxygen compound
- Organic nitrogen compound
- Hydrocarbon derivative
- Alcohol
- Organonitrogen compound
- Organooxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Exogenous |
|---|
| Cellular Locations | |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Appearance | White powder. |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available | | LogP | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | Deposition Date | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-052u-3000190000-97bd9ba4baababbac7f9 | 2017-09-01 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-03di-3301219000-42b2672f68cb064c4565 | 2017-10-06 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_2) - 70eV, Positive | Not Available | 2021-10-16 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_3) - 70eV, Positive | Not Available | 2021-10-16 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_4) - 70eV, Positive | Not Available | 2021-10-16 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_5) - 70eV, Positive | Not Available | 2021-10-16 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_1) - 70eV, Positive | Not Available | 2021-10-16 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_2) - 70eV, Positive | Not Available | 2021-10-16 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_3) - 70eV, Positive | Not Available | 2021-10-16 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_4) - 70eV, Positive | Not Available | 2021-10-16 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_5) - 70eV, Positive | Not Available | 2021-10-16 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_6) - 70eV, Positive | Not Available | 2021-10-16 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_7) - 70eV, Positive | Not Available | 2021-10-16 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_8) - 70eV, Positive | Not Available | 2021-10-16 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_9) - 70eV, Positive | Not Available | 2021-10-16 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_10) - 70eV, Positive | Not Available | 2021-10-16 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_3_1) - 70eV, Positive | Not Available | 2021-10-16 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_3_2) - 70eV, Positive | Not Available | 2021-10-16 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_3_3) - 70eV, Positive | Not Available | 2021-10-16 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_3_4) - 70eV, Positive | Not Available | 2021-10-16 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_3_5) - 70eV, Positive | Not Available | 2021-10-16 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_3_6) - 70eV, Positive | Not Available | 2021-10-16 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_3_7) - 70eV, Positive | Not Available | 2021-10-16 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_3_8) - 70eV, Positive | Not Available | 2021-10-16 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_3_9) - 70eV, Positive | Not Available | 2021-10-16 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00kr-0000091000-88a17f050534434d09e8 | 2016-06-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00my-0000090000-f40e4cbed40968d096cc | 2016-06-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-015i-2100960000-720410ac9cc84acb9cda | 2016-06-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-2000079000-79ae8bdff7c70c6272e5 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0f8i-3000192000-a3943e5f237df2c95298 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-5100940000-8f961fc229324898c9fb | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0000009000-87b11b40714eaca87637 | 2021-09-22 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0w29-0000197000-b7cee0092463e64db982 | 2021-09-22 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0kmi-0100091000-3ca4875fc89468926ad4 | 2021-09-22 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0000098000-6e6f765e43f3fb69e51a | 2021-09-22 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udr-0002093000-3fa96b188a5bdcc1a51c | 2021-09-22 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00r2-4121490000-3d169273a3c2564c172c | 2021-09-22 | View Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Oral, dermal, inhalation, and parenteral (contaminated drugs). (3) |
|---|
| Mechanism of Toxicity | Tremorgenic mycotoxins exert their toxic effects by interfering with neurotransmitter release, possibly by causing degeneration of nerve terminals. They are thought to inhibit gamma-aminobutyric acid (GABA) receptors, both pre- and postsynaptic, as well as inhibit transmitter breakdown at the GABA-T receptors. This would initially increase neurotransmitter levels, potentiating the GABA-induced chloride current, then lead to decreased levels of neurotransmitter in the synapse. (1) |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | No indication of carcinogenicity to humans (not listed by IARC). |
|---|
| Uses/Sources | Janthitrems are tremorgenic mycotoxins that have been found in the fungus Penicillium janthinellum. They may be found in contaminated cereal crops such as oats, barley, millet, corn and rice. (1, 2) |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Tremorgenic mycotoxins affect central nervous system activity. They cause a neurological disease of cattle known as "staggers syndrome". (1) |
|---|
| Symptoms | Tremorgenic mycotoxins affect central nervous system activity, inducing neurologic symptoms including mental confusion, paralysis, tremors, seizures, and death. They cause a neurological disease of cattle known as "staggers syndrome", which is characterized by muscle tremors, hyperexcitability, convulsions and ataxia. (1) |
|---|
| Treatment | To control severe tremors caused by tremorgenic mycotoxins, methocarbamol should be administered. Generalized seizures may be treated with diazepam followed by methocarbamol or a barbiturate such as pentobarbital sodium. Gastric lavage should be performed and activated charcoal administered to limit further absorption of toxins. (4) |
|---|
| Normal Concentrations |
|---|
| Not Available |
|---|
| Abnormal Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB30529 |
|---|
| PubChem Compound ID | 10348646 |
|---|
| ChEMBL ID | Not Available |
|---|
| ChemSpider ID | 8524104 |
|---|
| KEGG ID | Not Available |
|---|
| UniProt ID | Not Available |
|---|
| OMIM ID | |
|---|
| ChEBI ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| CTD ID | Not Available |
|---|
| Stitch ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| ACToR ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | - Selala MI, Daelemans F, Schepens PJ: Fungal tremorgens: the mechanism of action of single nitrogen containing toxins--a hypothesis. Drug Chem Toxicol. 1989 Sep-Dec;12(3-4):237-57. [2698801 ]
- Gallagher RT, Latch GC, Keogh RG: The janthitrems: fluorescent tremorgenic toxins produced by Penicillium janthinellum isolates from ryegrass pastures. Appl Environ Microbiol. 1980 Jan;39(1):272-3. [7356319 ]
- Peraica M, Domijan AM: Contamination of food with mycotoxins and human health. Arh Hig Rada Toksikol. 2001 Mar;52(1):23-35. [11370295 ]
- Prevention of stroke by antihypertensive drug treatment in older persons with isolated systolic hypertension. Final results of the Systolic Hypertension in the Elderly Program (SHEP). SHEP Cooperative Research Group. JAMA. 1991 Jun 26;265(24):3255-64. [2046107 ]
- Schell MM. Tremorgenic mycotoxin intoxication. Veterinary Medicine. 2000.
- Yannai, Shmuel. (2004) Dictionary of food compounds with CD-ROM: Additives, flavors, and ingredients. Boca Raton: Chapman & Hall/CRC.
|
|---|
| Gene Regulation |
|---|
| Up-Regulated Genes | Not Available |
|---|
| Down-Regulated Genes | Not Available |
|---|