| Record Information |
|---|
| Version | 2.0 |
|---|
| Creation Date | 2014-08-30 21:05:04 UTC |
|---|
| Update Date | 2014-12-24 20:26:52 UTC |
|---|
| Accession Number | T3D4564 |
|---|
| Identification |
|---|
| Common Name | Clobazam |
|---|
| Class | Small Molecule |
|---|
| Description | Clobazam belongs to the 1,5-benzodiazepine class of drugs and is expected to have a better side-effect profile compared to older 1,4-benzodiazepines. It has been marketed as an anxiolytic since 1975 and an anticonvulsant since 1984. The oral preparation was FDA approved on October 21, 2011. An oral suspension is expected to be available in 2013. |
|---|
| Compound Type | - Amide
- Amine
- Anticonvulsant
- Benzodiazepine
- Drug
- Organic Compound
- Organochloride
- Synthetic Compound
|
|---|
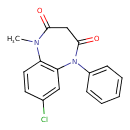
| Chemical Structure | |
|---|
| Synonyms | | Synonym | | 1-phenyl-5-methyl-8-chloro-1,2,4,5-tetrahydro-2,4-dioxo-3H-1,5-benzodiazepine | | 7-Chloro-1-methyl-5-phenyl-1,5-dihydro-benzo[b][1,4]diazepine-2,4-dione | | Aedon | | Castilium | | Clobam | | Clobamax | | Clobazamum | | Frisium | | Grifoclobam | | Mystan | | Noiafren | | Onfi | | Sederlona | | Urbanil | | Urbanol | | Urbanyl | | Venium |
|
|---|
| Chemical Formula | C16H13ClN2O2 |
|---|
| Average Molecular Mass | 300.740 g/mol |
|---|
| Monoisotopic Mass | 300.067 g/mol |
|---|
| CAS Registry Number | 22316-47-8 |
|---|
| IUPAC Name | 7-chloro-1-methyl-5-phenyl-2,3,4,5-tetrahydro-1H-1,5-benzodiazepine-2,4-dione |
|---|
| Traditional Name | clobazam |
|---|
| SMILES | CN1C2=C(C=C(Cl)C=C2)N(C2=CC=CC=C2)C(=O)CC1=O |
|---|
| InChI Identifier | InChI=1S/C16H13ClN2O2/c1-18-13-8-7-11(17)9-14(13)19(16(21)10-15(18)20)12-5-3-2-4-6-12/h2-9H,10H2,1H3 |
|---|
| InChI Key | InChIKey=CXOXHMZGEKVPMT-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as benzodiazepines. These are organic compounds containing a benzene ring fused to either isomers of diazepine(unsaturated seven-member heterocycle with two nitrogen atoms replacing two carbon atoms). |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Benzodiazepines |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Benzodiazepines |
|---|
| Alternative Parents | |
|---|
| Substituents | - Benzodiazepine
- Para-diazepine
- Aryl chloride
- Aryl halide
- Monocyclic benzene moiety
- 1,3-dicarbonyl compound
- Benzenoid
- Tertiary carboxylic acid amide
- Lactam
- Carboxamide group
- Carboxylic acid derivative
- Azacycle
- Organooxygen compound
- Organonitrogen compound
- Organochloride
- Organohalogen compound
- Carbonyl group
- Organic oxygen compound
- Organic nitrogen compound
- Hydrocarbon derivative
- Organic oxide
- Organopnictogen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Exogenous |
|---|
| Cellular Locations | |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | |
|---|
| Biological Roles | |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Appearance | White powder. |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | 180-182°C | | Boiling Point | Not Available | | Solubility | 188 mg/L | | LogP | 2.12 |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | Deposition Date | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-053r-0290000000-85c722aae16a6c4f1e1a | 2017-09-01 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QTOF , positive | splash10-0a4i-0090000000-3b46d60abcc8dfacf4d8 | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-0udi-0019000000-71512d7a18cf60430dd9 | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-0a4i-0092000000-e687a1e3760fb870dbc9 | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-0a4i-0090000000-d450ded1f22039f00b63 | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-0ab9-0190000000-e4211cbc2957a587e3d3 | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-05fr-0590000000-51f4fa0445c2d047ef6b | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-0g4j-1960000000-a89e1c91bfa7b974be48 | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-014j-4920000000-93c0877b3acf519d2b98 | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-016u-7900000000-bc29714be08b6ae4120b | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QFT , positive | splash10-0hi6-9600000000-7c6a3974d95a0015757e | 2017-09-14 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - -1V, Positive | splash10-0a4i-0090000000-f8bfd7fb18f6c2191b61 | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 15V, Positive | splash10-0udi-0019000000-71512d7a18cf60430dd9 | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 75V, Positive | splash10-05fr-0590000000-d54a49898cc15a62a5e6 | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 60V, Positive | splash10-0ab9-0190000000-55d5faa9832676ff3cbe | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 30V, Positive | splash10-0a4i-0092000000-ea34c0db78be09efb678 | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 45V, Positive | splash10-0a4i-0090000000-35ff57d7b80e2c98ae57 | 2021-09-20 | View Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - 90V, Positive | splash10-0g4j-1960000000-cf4e19e49420cb7d7a8f | 2021-09-20 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0009000000-c177e3ba787d31e6efa9 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udi-1019000000-ecf7835c598275ee85fb | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0f6x-9560000000-ac11ec0388fbae849904 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-0090000000-5aff96d7f16fb8885018 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0002-0090000000-38aef9b903ce595b395d | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0f9f-4490000000-02ff5e375a7cffbef887 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0009000000-d5f71cf6ca39e39e4e90 | 2021-09-24 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udi-0069000000-00f02d6bca08146095d7 | 2021-09-24 | View Spectrum | | MS | Mass Spectrum (Electron Ionization) | splash10-0pb9-6392000000-a76f30f353b3407df0c6 | 2014-09-20 | View Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | After oral administration of clobazam, it is almost completely absorbed (87% of dose). Bioavailability relative to solution was almost at 100%. Food does not affect absorption.
Tmax = 1-3 hours. |
|---|
| Mechanism of Toxicity | Clobazam binds at distinct binding sites associated with the chloride ionopore at the post-synaptic GABA receptor. These GABA receptors are in various locations in the CNS (limbic, reticular formation) and clobazam increases the duration of time for which the chloride ionopore is open. As a result, hyper polarization and stabilization of the membrane occur as the post-synaptic inhibitory effect of GABA is enhanced. |
|---|
| Metabolism | Clobazam is extensively metabolized in the liver via N-demethylation and hydroxylation. Clobazam has two major metabolites: N-desmethylclobazam (norclobazam) and 4'-hydroxyclobazam, the former of which is active. Norclobazam is one-fourth the potency of clobazam. The main enzyme that facilitates the process of N-demethylation is CYP3A4, and to a lesser extent by CYP2C19 and CYP2B6. Norclobazam itself is also metabolized via hydroxylation, primarily by CYP2C19. The formation of 4'-hydroxyclobazam is facilitated by CYP2C18 and CYP2C19. A factor in determining extent of metabolism is the genetic profile of the individual patient as CYP2C19 is a polymorphic enzyme.
Route of Elimination: Clobazam is eliminated via the urine (~94%) as metabolites.
Half Life: The mean elimination half life of an oral dose of clobazam 40 mg is 32 hours. It's main metabolite, norclobazam, as a half life of 57 hours. The half life in adult patients with epilepsy are higher than those that are healthy. |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | No indication of carcinogenicity to humans (not listed by IARC). |
|---|
| Uses/Sources | For treatment and management of epilepsy and seizures associated with Lennox-Gastaut syndrome, a difficult-to-treat form of childhood epilepsy. |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | The most common adverse effects include somnolence, pyrexia, upper respiratory tract infection, and lethargy. |
|---|
| Treatment | General supportive measures should be employed, along with intravenous fluids, and an adequate airway maintained. Hypotension may be combated by the use of norepinephrine or metaraminol. Dialysis is of limited value. Flumazenil (Anexate) is a competitive benzodiazepine receptor antagonist that can be used as an antidote for benzodiazepine overdose. In particular, flumazenil is very effective at reversing the CNS depression associated with benzodiazepines but is less effective at reversing respiratory depression. Its use, however, is controversial as it has numerous contraindications. It is contraindicated in patients who are on long-term benzodiazepines, those who have ingested a substance that lowers the seizure threshold, or in patients who have tachycardia or a history of seizures. As a general rule, medical observation and supportive care are the mainstay of treatment of benzodiazepine overdose. Although benzodiazepines are absorbed by activated charcoal, gastric decontamination with activated charcoal is not beneficial in pure benzodiazepine overdose as the risk of adverse effects often outweigh any potential benefit from the procedure. It is recommended only if benzodiazepines have been taken in combination with other drugs that may benefit from decontamination. Gastric lavage (stomach pumping) or whole bowel irrigation are also not recommended. |
|---|
| Normal Concentrations |
|---|
| Not Available |
|---|
| Abnormal Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB00349 |
|---|
| HMDB ID | Not Available |
|---|
| PubChem Compound ID | 2789 |
|---|
| ChEMBL ID | CHEMBL70418 |
|---|
| ChemSpider ID | 2687 |
|---|
| KEGG ID | Not Available |
|---|
| UniProt ID | Not Available |
|---|
| OMIM ID | |
|---|
| ChEBI ID | 31413 |
|---|
| BioCyc ID | Not Available |
|---|
| CTD ID | Not Available |
|---|
| Stitch ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| ACToR ID | Not Available |
|---|
| Wikipedia Link | Clobazam |
|---|
| References |
|---|
| Synthesis Reference | Hauptmann, K.H., Weber, K.-H., Zeile, K., Danneberg, P. and Giesemann, R.; South African
Patent 68/0803; February 7,1968; assigned to Boehringer lngelheim GmbH, Germany. |
|---|
| MSDS | T3D4564.pdf |
|---|
| General References | - Freche C: [Study of an anxiolytic, clobazam, in otorhinolaryngology in psychosomatic pharyngeal manifestations]. Sem Hop Ther. 1975 Apr;51(4):261-3. [5777 ]
- Clobazam in treatment of refractory epilepsy: the Canadian experience. A retrospective study. Canadian Clobazam Cooperative Group. Epilepsia. 1991 May-Jun;32(3):407-16. [2044502 ]
- Wildin JD, Pleuvry BJ, Mawer GE, Onon T, Millington L: Respiratory and sedative effects of clobazam and clonazepam in volunteers. Br J Clin Pharmacol. 1990 Feb;29(2):169-77. [2106335 ]
- Kilpatrick C, Bury R, Fullinfaw R, Moulds R: Clobazam in the treatment of epilepsy. Clin Exp Neurol. 1987;23:139-44. [3117456 ]
- Giarratano M, Standley K, Benbadis SR: Clobazam for treatment of epilepsy. Expert Opin Pharmacother. 2012 Feb;13(2):227-33. doi: 10.1517/14656566.2012.647686. Epub 2012 Jan 13. [22242724 ]
- Yang LP, Scott LJ: Clobazam : in patients with Lennox-Gastaut syndrome. CNS Drugs. 2012 Nov;26(11):983-91. doi: 10.1007/s40263-012-0007-0. [23034582 ]
- Walzer M, Bekersky I, Blum RA, Tolbert D: Pharmacokinetic drug interactions between clobazam and drugs metabolized by cytochrome P450 isoenzymes. Pharmacotherapy. 2012 Apr;32(4):340-53. doi: 10.1002/j.1875-9114.2012.01028.x. Epub 2012 Mar 15. [22422635 ]
|
|---|
| Gene Regulation |
|---|
| Up-Regulated Genes | Not Available |
|---|
| Down-Regulated Genes | Not Available |
|---|