Propachlor (T3D1090)
| Record Information | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 2.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Creation Date | 2009-06-18 21:54:34 UTC | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 2014-12-24 20:23:07 UTC | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Accession Number | T3D1090 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Common Name | Propachlor | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Small Molecule | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | Propachlor is widely used as a selective herbicide worldwide in corn, soybean and other crop cultures. Elevated concentrations of these herbicides and their degradation products have been detected in surface and groundwater. Propachlor is a pre-emergence herbicide, effective against annual grasses and certain broad-leaved weeds. It is a compound of the family of chloroacetanilides. (1, 8) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Compound Type |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
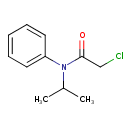
| Chemical Structure | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula | C11H14ClNO | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Mass | 211.688 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Mass | 211.076 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS Registry Number | 1918-16-7 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name | 2-chloro-N-phenyl-N-(propan-2-yl)acetamide | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional Name | propachlor | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES | CC(C)N(C(=O)CCl)C1=CC=CC=C1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Identifier | InChI=1S/C11H14ClNO/c1-9(2)13(11(14)8-12)10-6-4-3-5-7-10/h3-7,9H,8H2,1-2H3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key | InChIKey=MFOUDYKPLGXPGO-UHFFFAOYSA-N | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | belongs to the class of organic compounds known as anilides. These are organic heterocyclic compounds derived from oxoacids RkE(=O)l(OH)m (l not 0) by replacing an OH group by the NHPh group or derivative formed by ring substitution. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Organic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Benzenoids | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Benzene and substituted derivatives | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Anilides | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Anilides | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Aromatic homomonocyclic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Status | Detected and Not Quantified | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Origin | Exogenous | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biofluid Locations | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tissue Locations | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Applications | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Roles | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Roles | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State | Solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | Tan colored solid (8). | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Profile | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Route of Exposure | Oral (8) ; inhalation (8) ; dermal (8) ; dermal (8). | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism of Toxicity | Propachlor poisoning causes an induction of microsomal hepatic UDPGT activity, which produces increased clearance of thyroid hormone, T4. Decreased levels of T4 would result in increased levels of thyroid stimulating hormone (TSH). Increased levels of TSH would result in the hyperplastic and eventually tumorigenic response of the thyroid. Propachlor metabolites also bind to nAChRs in the nervous system and cause endocrine disruption in humans by binding to and inhibiting the estrogen receptor. (3, 2, 6) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolism | Propachlor can be absorbed in mammals through the respiratory and gastrointestinal tracts as well as through the skin. It does not accumulate in the body, since it is rapidly metabolized through the mercapturic acid pathway (MAP). Cysteine conjugates are formed by glutathione conjugation and this conjugate has been proposed as an intermediate in the metabolic formation of mercapturic acid conjugation. It has been shown that a cysteine conjugate of propachlor is the source of sulfur in methylsulfonyl- containing metabolites, but that the carbon in the methylsulfonyl group does not come from the cysteine moiety. Propachlor is conjugated firstly with glutathione and the reaction is mediated by glutathione transferases. The glutathione conjugation provides a means for inactivation of reactive electrophiles. Glutathione conjugates have the required physico-chemical properties for biliary excretion and will generally be present, together with their catabolites cysteinyl-glycine, cysteine and N-acetylcysteine-mercapturic acid, in relatively high concentrations in the bile. After excretion with the bile, they are metabolized in the intestine where the C-S lyase present cleaves the cysteine conjugate, allowing further metabolism of sulfur to a methylsulfonyl-containing moiety. Eleven urinary metabolites, six of which are 2-methylsulfonylacetanalides have been isolated. The major fecal metabolite is identified as the cysteine conjugate. (8, 9, 10) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Values | LD50: 950-2176 mg/kg (Oral, Rat) (8) LC50: 3580 mg/m3 (Inhalation, Rat) (9) LD50: 380 mg/kg (Dermal, Rabbit) (9) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lethal Dose | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carcinogenicity (IARC Classification) | Not listed by IARC. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Uses/Sources | Propachlor is used on a variety of food plants, including onions, field corn, hybrid seed, silage corn, sweet corn, grain sorghum, green peas, soybeans, flax, pumpkin, and strawberries. (8) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Minimum Risk Level | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Health Effects | Eye exposure may cause corneal damage. Erythematopapular contact eczema can follow exposure to propachlor. (8, 9) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symptoms | Irritation and/or severe burns of the skin, eye, esophagus or gastrointestinal tract, and the respiratory tract, depending on the route of exposure. (4) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Treatment | Consider gastric lavage, as well was dilution with milk or water after ingestion. Administer charcoal as a slurry following ingestion; however, activated charcoal should not be given to patients ingesting strong acidic or basic caustic chemicals. In case of inhalation, move patient to fresh air. Monitor for respiratory distress. If cough or difficulty breathing develops, evaluate for respiratory tract irritation, bronchitis, or pneumonitis. Administer oxygen and assist ventilation as required. Treat bronchospasm with inhaled beta2 agonist and oral or parenteral corticosteroids. Irrigate exposed eyes with copious amounts of room temperature water for at least 15 minutes. Following dermal exposure, remove contaminated clothing and wash exposed area thoroughly with soap and water. Treat dermal irritation or burns with standard topical therapy. Patients developing dermal hypersensitivity reactions may require treatment with systemic or topical corticosteroids or antihistamines. Administer symptomatic treatment as necessary. (4) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Normal Concentrations | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abnormal Concentrations | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| DrugBank ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HMDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PubChem Compound ID | 4931 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ChEMBL ID | CHEMBL1394829 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ChemSpider ID | 4762 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| KEGG ID | C18759 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| UniProt ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OMIM ID | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ChEBI ID | 19503 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BioCyc ID | ALPHA-NAPHTHALENEACETAMIDE | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CTD ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stitch ID | Propachlor | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ACToR ID | 6518 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikipedia Link | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MSDS | T3D1090.pdf | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General References |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gene Regulation | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Up-Regulated Genes | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Down-Regulated Genes | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Targets
- General Function:
- Receptor binding
- Specific Function:
- Ionotropic receptor with a probable role in the modulation of auditory stimuli. Agonist binding may induce an extensive change in conformation that affects all subunits and leads to opening of an ion-conducting channel across the plasma membrane. The channel is permeable to a range of divalent cations including calcium, the influx of which may activate a potassium current which hyperpolarizes the cell membrane. In the ear, this may lead to a reduction in basilar membrane motion, altering the activity of auditory nerve fibers and reducing the range of dynamic hearing. This may protect against acoustic trauma.
- Gene Name:
- CHRNA10
- Uniprot ID:
- Q9GZZ6
- Molecular Weight:
- 49704.295 Da
References
- Belyaeva OV, Kedishvili NY: Comparative genomic and phylogenetic analysis of short-chain dehydrogenases/reductases with dual retinol/sterol substrate specificity. Genomics. 2006 Dec;88(6):820-30. Epub 2006 Jul 24. [16860536 ]
- Casarett LJ, Klaassen CD, and Watkins JB (2003). Casarett and Doull's essentials of toxicology. New York: McGraw-Hill/Medical Pub. Div.
- EPA Office of Pesticide Programs (2001). Implementation of the Determinations of a Common Mechanism of Toxicity for N-Methyl Carbamate Pesticides and for Certain Chloroacetaide Pesticides.
- General Function:
- Ligand-gated ion channel activity
- Specific Function:
- After binding acetylcholine, the AChR responds by an extensive change in conformation that affects all subunits and leads to opening of an ion-conducting channel across the plasma membrane.
- Gene Name:
- CHRNA3
- Uniprot ID:
- P32297
- Molecular Weight:
- 57479.54 Da
References
- Belyaeva OV, Kedishvili NY: Comparative genomic and phylogenetic analysis of short-chain dehydrogenases/reductases with dual retinol/sterol substrate specificity. Genomics. 2006 Dec;88(6):820-30. Epub 2006 Jul 24. [16860536 ]
- Casarett LJ, Klaassen CD, and Watkins JB (2003). Casarett and Doull's essentials of toxicology. New York: McGraw-Hill/Medical Pub. Div.
- EPA Office of Pesticide Programs (2001). Implementation of the Determinations of a Common Mechanism of Toxicity for N-Methyl Carbamate Pesticides and for Certain Chloroacetaide Pesticides.
- General Function:
- Ligand-gated ion channel activity
- Specific Function:
- After binding acetylcholine, the AChR responds by an extensive change in conformation that affects all subunits and leads to opening of an ion-conducting channel across the plasma membrane permeable to sodium ions.
- Gene Name:
- CHRNA4
- Uniprot ID:
- P43681
- Molecular Weight:
- 69956.47 Da
References
- Belyaeva OV, Kedishvili NY: Comparative genomic and phylogenetic analysis of short-chain dehydrogenases/reductases with dual retinol/sterol substrate specificity. Genomics. 2006 Dec;88(6):820-30. Epub 2006 Jul 24. [16860536 ]
- Casarett LJ, Klaassen CD, and Watkins JB (2003). Casarett and Doull's essentials of toxicology. New York: McGraw-Hill/Medical Pub. Div.
- EPA Office of Pesticide Programs (2001). Implementation of the Determinations of a Common Mechanism of Toxicity for N-Methyl Carbamate Pesticides and for Certain Chloroacetaide Pesticides.
- General Function:
- Ligand-gated ion channel activity
- Specific Function:
- After binding acetylcholine, the AChR responds by an extensive change in conformation that affects all subunits and leads to opening of an ion-conducting channel across the plasma membrane.
- Gene Name:
- CHRNA5
- Uniprot ID:
- P30532
- Molecular Weight:
- 53053.965 Da
References
- Belyaeva OV, Kedishvili NY: Comparative genomic and phylogenetic analysis of short-chain dehydrogenases/reductases with dual retinol/sterol substrate specificity. Genomics. 2006 Dec;88(6):820-30. Epub 2006 Jul 24. [16860536 ]
- Casarett LJ, Klaassen CD, and Watkins JB (2003). Casarett and Doull's essentials of toxicology. New York: McGraw-Hill/Medical Pub. Div.
- EPA Office of Pesticide Programs (2001). Implementation of the Determinations of a Common Mechanism of Toxicity for N-Methyl Carbamate Pesticides and for Certain Chloroacetaide Pesticides.
- General Function:
- Acetylcholine-activated cation-selective channel activity
- Specific Function:
- After binding acetylcholine, the AChR responds by an extensive change in conformation that affects all subunits and leads to opening of an ion-conducting channel across the plasma membrane.
- Gene Name:
- CHRNA6
- Uniprot ID:
- Q15825
- Molecular Weight:
- 56897.745 Da
References
- Belyaeva OV, Kedishvili NY: Comparative genomic and phylogenetic analysis of short-chain dehydrogenases/reductases with dual retinol/sterol substrate specificity. Genomics. 2006 Dec;88(6):820-30. Epub 2006 Jul 24. [16860536 ]
- Casarett LJ, Klaassen CD, and Watkins JB (2003). Casarett and Doull's essentials of toxicology. New York: McGraw-Hill/Medical Pub. Div.
- EPA Office of Pesticide Programs (2001). Implementation of the Determinations of a Common Mechanism of Toxicity for N-Methyl Carbamate Pesticides and for Certain Chloroacetaide Pesticides.
- General Function:
- Toxic substance binding
- Specific Function:
- After binding acetylcholine, the AChR responds by an extensive change in conformation that affects all subunits and leads to opening of an ion-conducting channel across the plasma membrane. The channel is blocked by alpha-bungarotoxin.
- Gene Name:
- CHRNA7
- Uniprot ID:
- P36544
- Molecular Weight:
- 56448.925 Da
References
- Belyaeva OV, Kedishvili NY: Comparative genomic and phylogenetic analysis of short-chain dehydrogenases/reductases with dual retinol/sterol substrate specificity. Genomics. 2006 Dec;88(6):820-30. Epub 2006 Jul 24. [16860536 ]
- Casarett LJ, Klaassen CD, and Watkins JB (2003). Casarett and Doull's essentials of toxicology. New York: McGraw-Hill/Medical Pub. Div.
- EPA Office of Pesticide Programs (2001). Implementation of the Determinations of a Common Mechanism of Toxicity for N-Methyl Carbamate Pesticides and for Certain Chloroacetaide Pesticides.
- General Function:
- Calcium channel activity
- Specific Function:
- Ionotropic receptor with a probable role in the modulation of auditory stimuli. Agonist binding induces a conformation change that leads to the opening of an ion-conducting channel across the plasma membrane (PubMed:11752216, PubMed:25282151). The channel is permeable to a range of divalent cations including calcium, the influx of which may activate a potassium current which hyperpolarizes the cell membrane (PubMed:11752216, PubMed:25282151). In the ear, this may lead to a reduction in basilar membrane motion, altering the activity of auditory nerve fibers and reducing the range of dynamic hearing. This may protect against acoustic trauma. May also regulate keratinocyte adhesion (PubMed:11021840).
- Gene Name:
- CHRNA9
- Uniprot ID:
- Q9UGM1
- Molecular Weight:
- 54806.63 Da
References
- Belyaeva OV, Kedishvili NY: Comparative genomic and phylogenetic analysis of short-chain dehydrogenases/reductases with dual retinol/sterol substrate specificity. Genomics. 2006 Dec;88(6):820-30. Epub 2006 Jul 24. [16860536 ]
- Casarett LJ, Klaassen CD, and Watkins JB (2003). Casarett and Doull's essentials of toxicology. New York: McGraw-Hill/Medical Pub. Div.
- EPA Office of Pesticide Programs (2001). Implementation of the Determinations of a Common Mechanism of Toxicity for N-Methyl Carbamate Pesticides and for Certain Chloroacetaide Pesticides.
- General Function:
- Ligand-gated ion channel activity
- Specific Function:
- After binding acetylcholine, the AChR responds by an extensive change in conformation that affects all subunits and leads to opening of an ion-conducting channel across the plasma membrane permeable to sodiun ions.
- Gene Name:
- CHRNB2
- Uniprot ID:
- P17787
- Molecular Weight:
- 57018.575 Da
References
- Belyaeva OV, Kedishvili NY: Comparative genomic and phylogenetic analysis of short-chain dehydrogenases/reductases with dual retinol/sterol substrate specificity. Genomics. 2006 Dec;88(6):820-30. Epub 2006 Jul 24. [16860536 ]
- Casarett LJ, Klaassen CD, and Watkins JB (2003). Casarett and Doull's essentials of toxicology. New York: McGraw-Hill/Medical Pub. Div.
- EPA Office of Pesticide Programs (2001). Implementation of the Determinations of a Common Mechanism of Toxicity for N-Methyl Carbamate Pesticides and for Certain Chloroacetaide Pesticides.
- General Function:
- Drug binding
- Specific Function:
- After binding acetylcholine, the AChR responds by an extensive change in conformation that affects all subunits and leads to opening of an ion-conducting channel across the plasma membrane.
- Gene Name:
- CHRNB3
- Uniprot ID:
- Q05901
- Molecular Weight:
- 52728.215 Da
References
- Belyaeva OV, Kedishvili NY: Comparative genomic and phylogenetic analysis of short-chain dehydrogenases/reductases with dual retinol/sterol substrate specificity. Genomics. 2006 Dec;88(6):820-30. Epub 2006 Jul 24. [16860536 ]
- Casarett LJ, Klaassen CD, and Watkins JB (2003). Casarett and Doull's essentials of toxicology. New York: McGraw-Hill/Medical Pub. Div.
- EPA Office of Pesticide Programs (2001). Implementation of the Determinations of a Common Mechanism of Toxicity for N-Methyl Carbamate Pesticides and for Certain Chloroacetaide Pesticides.
- General Function:
- Ligand-gated ion channel activity
- Specific Function:
- After binding acetylcholine, the AChR responds by an extensive change in conformation that affects all subunits and leads to opening of an ion-conducting channel across the plasma membrane.
- Gene Name:
- CHRNB4
- Uniprot ID:
- P30926
- Molecular Weight:
- 56378.985 Da
References
- Belyaeva OV, Kedishvili NY: Comparative genomic and phylogenetic analysis of short-chain dehydrogenases/reductases with dual retinol/sterol substrate specificity. Genomics. 2006 Dec;88(6):820-30. Epub 2006 Jul 24. [16860536 ]
- Casarett LJ, Klaassen CD, and Watkins JB (2003). Casarett and Doull's essentials of toxicology. New York: McGraw-Hill/Medical Pub. Div.
- EPA Office of Pesticide Programs (2001). Implementation of the Determinations of a Common Mechanism of Toxicity for N-Methyl Carbamate Pesticides and for Certain Chloroacetaide Pesticides.
- General Function:
- Zinc ion binding
- Specific Function:
- Nuclear hormone receptor. The steroid hormones and their receptors are involved in the regulation of eukaryotic gene expression and affect cellular proliferation and differentiation in target tissues. Ligand-dependent nuclear transactivation involves either direct homodimer binding to a palindromic estrogen response element (ERE) sequence or association with other DNA-binding transcription factors, such as AP-1/c-Jun, c-Fos, ATF-2, Sp1 and Sp3, to mediate ERE-independent signaling. Ligand binding induces a conformational change allowing subsequent or combinatorial association with multiprotein coactivator complexes through LXXLL motifs of their respective components. Mutual transrepression occurs between the estrogen receptor (ER) and NF-kappa-B in a cell-type specific manner. Decreases NF-kappa-B DNA-binding activity and inhibits NF-kappa-B-mediated transcription from the IL6 promoter and displace RELA/p65 and associated coregulators from the promoter. Recruited to the NF-kappa-B response element of the CCL2 and IL8 promoters and can displace CREBBP. Present with NF-kappa-B components RELA/p65 and NFKB1/p50 on ERE sequences. Can also act synergistically with NF-kappa-B to activate transcription involving respective recruitment adjacent response elements; the function involves CREBBP. Can activate the transcriptional activity of TFF1. Also mediates membrane-initiated estrogen signaling involving various kinase cascades. Isoform 3 is involved in activation of NOS3 and endothelial nitric oxide production. Isoforms lacking one or several functional domains are thought to modulate transcriptional activity by competitive ligand or DNA binding and/or heterodimerization with the full length receptor. Essential for MTA1-mediated transcriptional regulation of BRCA1 and BCAS3. Isoform 3 can bind to ERE and inhibit isoform 1.
- Gene Name:
- ESR1
- Uniprot ID:
- P03372
- Molecular Weight:
- 66215.45 Da
References
- Taccone-Gallucci M, Manca-di-Villahermosa S, Battistini L, Stuffler RG, Tedesco M, Maccarrone M: N-3 PUFAs reduce oxidative stress in ESRD patients on maintenance HD by inhibiting 5-lipoxygenase activity. Kidney Int. 2006 Apr;69(8):1450-4. [16531984 ]
- Luft S, Milki E, Glustrom E, Ampiah-Bonney R, O'Hara P. Binding of Organochloride and Pyrethroid Pesticides To Estrogen Receptors α and β: A Fluorescence Polarization Assay. Biophysical Journal 2009;96(3):444a.
- General Function:
- Zinc ion binding
- Specific Function:
- Nuclear hormone receptor. Binds estrogens with an affinity similar to that of ESR1, and activates expression of reporter genes containing estrogen response elements (ERE) in an estrogen-dependent manner (PubMed:20074560). Isoform beta-cx lacks ligand binding ability and has no or only very low ere binding activity resulting in the loss of ligand-dependent transactivation ability. DNA-binding by ESR1 and ESR2 is rapidly lost at 37 degrees Celsius in the absence of ligand while in the presence of 17 beta-estradiol and 4-hydroxy-tamoxifen loss in DNA-binding at elevated temperature is more gradual.
- Gene Name:
- ESR2
- Uniprot ID:
- Q92731
- Molecular Weight:
- 59215.765 Da
References
- Taccone-Gallucci M, Manca-di-Villahermosa S, Battistini L, Stuffler RG, Tedesco M, Maccarrone M: N-3 PUFAs reduce oxidative stress in ESRD patients on maintenance HD by inhibiting 5-lipoxygenase activity. Kidney Int. 2006 Apr;69(8):1450-4. [16531984 ]
- Luft S, Milki E, Glustrom E, Ampiah-Bonney R, O'Hara P. Binding of Organochloride and Pyrethroid Pesticides To Estrogen Receptors α and β: A Fluorescence Polarization Assay. Biophysical Journal 2009;96(3):444a.
- General Function:
- Temperature-gated cation channel activity
- Specific Function:
- Receptor-activated non-selective cation channel involved in detection of pain and possibly also in cold perception and inner ear function (PubMed:25389312, PubMed:25855297). Has a central role in the pain response to endogenous inflammatory mediators and to a diverse array of volatile irritants, such as mustard oil, cinnamaldehyde, garlic and acrolein, an irritant from tears gas and vehicule exhaust fumes (PubMed:25389312, PubMed:20547126). Is also activated by menthol (in vitro)(PubMed:25389312). Acts also as a ionotropic cannabinoid receptor by being activated by delta(9)-tetrahydrocannabinol (THC), the psychoactive component of marijuana (PubMed:25389312). May be a component for the mechanosensitive transduction channel of hair cells in inner ear, thereby participating in the perception of sounds. Probably operated by a phosphatidylinositol second messenger system (By similarity).
- Gene Name:
- TRPA1
- Uniprot ID:
- O75762
- Molecular Weight:
- 127499.88 Da
References
- Nilius B, Prenen J, Owsianik G: Irritating channels: the case of TRPA1. J Physiol. 2011 Apr 1;589(Pt 7):1543-9. doi: 10.1113/jphysiol.2010.200717. Epub 2010 Nov 15. [21078588 ]
- General Function:
- Zinc ion binding
- Specific Function:
- Nuclear receptor that binds peroxisome proliferators such as hypolipidemic drugs and fatty acids. Once activated by a ligand, the nuclear receptor binds to DNA specific PPAR response elements (PPRE) and modulates the transcription of its target genes, such as acyl-CoA oxidase. It therefore controls the peroxisomal beta-oxidation pathway of fatty acids. Key regulator of adipocyte differentiation and glucose homeostasis. ARF6 acts as a key regulator of the tissue-specific adipocyte P2 (aP2) enhancer. Acts as a critical regulator of gut homeostasis by suppressing NF-kappa-B-mediated proinflammatory responses. Plays a role in the regulation of cardiovascular circadian rhythms by regulating the transcription of ARNTL/BMAL1 in the blood vessels (By similarity).
- Gene Name:
- PPARG
- Uniprot ID:
- P37231
- Molecular Weight:
- 57619.58 Da
Binding/Activity Constants
| Type | Value | Assay Type | Assay Source |
|---|---|---|---|
| AC50 | 2.75 uM | ATG_PPARg_TRANS | Attagene |
References
- Sipes NS, Martin MT, Kothiya P, Reif DM, Judson RS, Richard AM, Houck KA, Dix DJ, Kavlock RJ, Knudsen TB: Profiling 976 ToxCast chemicals across 331 enzymatic and receptor signaling assays. Chem Res Toxicol. 2013 Jun 17;26(6):878-95. doi: 10.1021/tx400021f. Epub 2013 May 16. [23611293 ]
- General Function:
- Zinc ion binding
- Specific Function:
- Steroid hormone receptors are ligand-activated transcription factors that regulate eukaryotic gene expression and affect cellular proliferation and differentiation in target tissues. Transcription factor activity is modulated by bound coactivator and corepressor proteins. Transcription activation is down-regulated by NR0B2. Activated, but not phosphorylated, by HIPK3 and ZIPK/DAPK3.
- Gene Name:
- AR
- Uniprot ID:
- P10275
- Molecular Weight:
- 98987.9 Da
Binding/Activity Constants
| Type | Value | Assay Type | Assay Source |
|---|---|---|---|
| AC50 | 3.99 uM | Tox21_AR_BLA_Antagonist_ratio | Tox21/NCGC |
References
- Sipes NS, Martin MT, Kothiya P, Reif DM, Judson RS, Richard AM, Houck KA, Dix DJ, Kavlock RJ, Knudsen TB: Profiling 976 ToxCast chemicals across 331 enzymatic and receptor signaling assays. Chem Res Toxicol. 2013 Jun 17;26(6):878-95. doi: 10.1021/tx400021f. Epub 2013 May 16. [23611293 ]
- General Function:
- Cholesterol binding
- Specific Function:
- Can bind protoporphyrin IX and may play a role in the transport of porphyrins and heme (By similarity). Promotes the transport of cholesterol across mitochondrial membranes and may play a role in lipid metabolism (PubMed:24814875), but its precise physiological role is controversial. It is apparently not required for steroid hormone biosynthesis. Was initially identified as peripheral-type benzodiazepine receptor; can also bind isoquinoline carboxamides (PubMed:1847678).
- Gene Name:
- TSPO
- Uniprot ID:
- P30536
- Molecular Weight:
- 18827.81 Da
Binding/Activity Constants
| Type | Value | Assay Type | Assay Source |
|---|---|---|---|
| AC50 | 6.62 uM | NVS_MP_hPBR | Novascreen |
References
- Sipes NS, Martin MT, Kothiya P, Reif DM, Judson RS, Richard AM, Houck KA, Dix DJ, Kavlock RJ, Knudsen TB: Profiling 976 ToxCast chemicals across 331 enzymatic and receptor signaling assays. Chem Res Toxicol. 2013 Jun 17;26(6):878-95. doi: 10.1021/tx400021f. Epub 2013 May 16. [23611293 ]
- General Function:
- Oxygen binding
- Specific Function:
- Catalyzes the formation of aromatic C18 estrogens from C19 androgens.
- Gene Name:
- CYP19A1
- Uniprot ID:
- P11511
- Molecular Weight:
- 57882.48 Da
Binding/Activity Constants
| Type | Value | Assay Type | Assay Source |
|---|---|---|---|
| AC50 | 8.49 uM | Tox21_Aromatase_Inhibition | Tox21/NCGC |
References
- Sipes NS, Martin MT, Kothiya P, Reif DM, Judson RS, Richard AM, Houck KA, Dix DJ, Kavlock RJ, Knudsen TB: Profiling 976 ToxCast chemicals across 331 enzymatic and receptor signaling assays. Chem Res Toxicol. 2013 Jun 17;26(6):878-95. doi: 10.1021/tx400021f. Epub 2013 May 16. [23611293 ]